Homolytic cleavage, or homolysis, is a chemical reaction in which a covalent bond between two atoms is broken equally, and each atom retains one of the two electrons that form the bond. This creates two species (similar or dissimilar), each with an unpaired electron, known as the radicals. The radical electron is denoted with a dot (.) over the atoms’ symbol.
For example, the homolytic bond cleavage of chlorine (Cl2) and HBr is shown below, resulting in similar and dissimilar radicals.
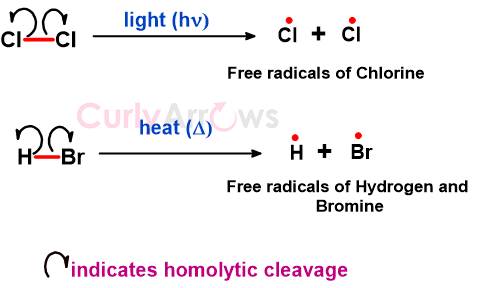
The homolytic cleavage is indicated with a half-headed (fishhook) arrow with the arrow base on the bond and arrowhead on the receiver atom of the bond electron.
The homolytic cleavage is initiated by heat (∆) or light (hν), which provides enough energy to break the bond and separate the two atoms. The resulting radicals are highly reactive and can lead to different chemical reactions like polymerization, halogenation, oxidation-reduction, etc., therefore integral for various biological, industrial, and environmental processes.
Related Reading: Free Radical, Introduction, Structure and Types
