London Dispersion, a type of Vander Waal Force, is the weakest of the three types, yet, it is the only one universally present in all the atoms and molecules.
In most cases, it is present in addition to the other forces; however, it is exclusively observed in nonpolar atoms and molecules devoid of any functional groups and, therefore, possess no other overpowering interactions. Examples are He, CH4, I2, C(CH3)4, etc.
Despite being the weakest, London dispersion force plays a crucial role in altering the state of atoms and molecules from gas to liquid and solid, affects the physical properties of the melting and boiling points, even extending its utility biologically in drug-receptor interactions, and more.
It occurs due to the constant, random electron fluctuations that redistribute the electron density leading to a moment where one area is more electron-rich and the other electron-deficient, invariably forming temporary poles. These poles induce pole formation in nearby atoms and molecules, dispersing the effect. They begin to align and collectively interact by an attraction, so such an attractive interaction is also known as an instantaneous dipole-induced dipole.
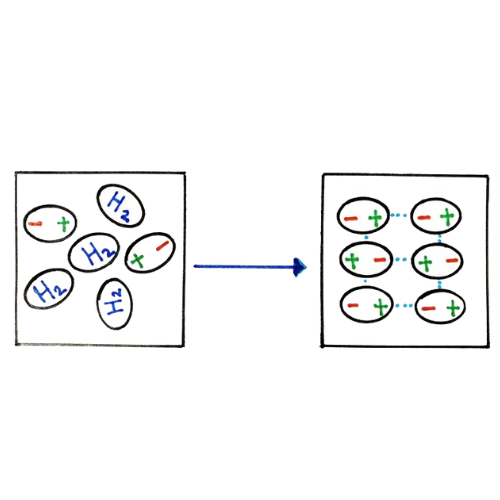
The strength of London Dispersion is greatly affected by the atom's or the molecule's shape and size, where the effect is predominant in bigger atoms and linear molecules.
