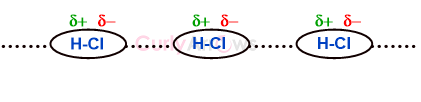
A permanent dipole is an inherent feature of the molecule due to the nature of the participating atoms forming the two-electron covalent bond.
Some atoms withdraw more of the bond electrons towards themselves, increasing their electron density and leaving the other end electron deficient. This unequal electron charge distribution creates positive and negative poles (denoted with δ+, δ- signs), causing the molecule to have permanent poles or a dipole.

A molecule with a permanent dipole is called a polar molecule.
Examples where the polar bonds create polar molecules are- H-F, H-O-H (H-O), H3C-OH (C-O), and H3C-NH2 (C-N).
The dipoles can electrostatically attract, resulting in a type of intermolecular force called the Dipole-Dipole or Keesom Interaction.
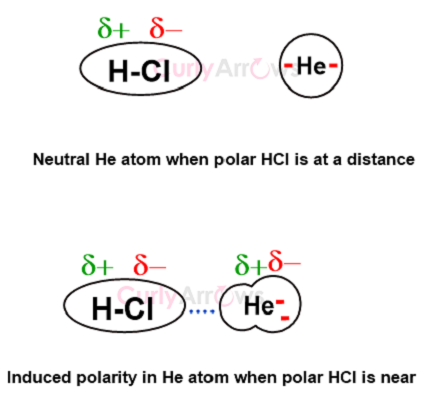
These permanent dipoles can also induce polarity in otherwise nonpolar molecules in Dipole-Induced Dipole or Debye interactions.
However, there are instances where the molecule's geometry is responsible for non-polarity in otherwise polar molecules containing polar bonds. Example, CO2, BF3, SO3, CCl4, etc.
