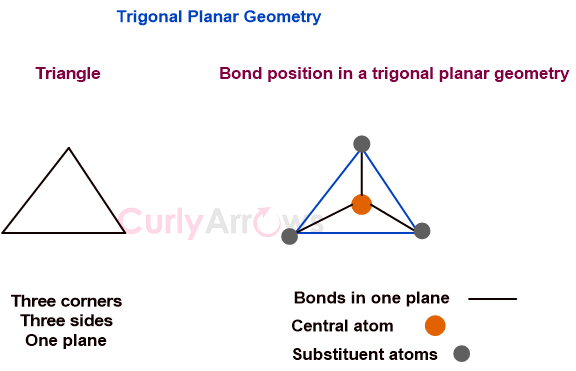
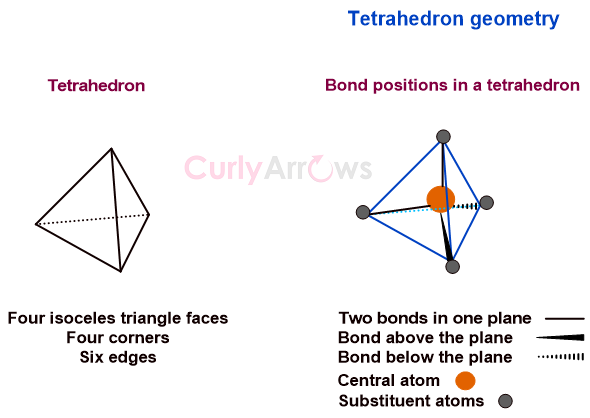
Definition of Valence Electrons
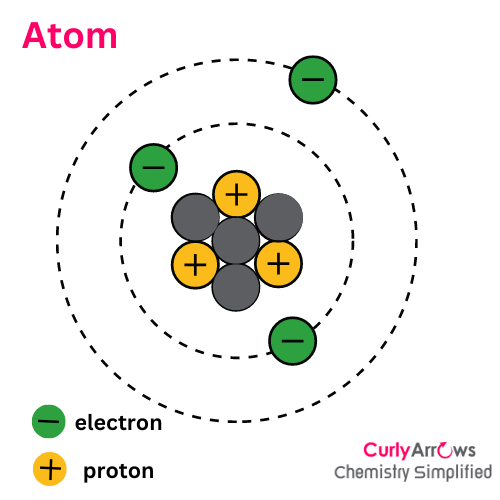
The outermost electrons of an atom that are mostly involved in bonding reactions are called valence electrons. These electrons are farthest from the nucleus and have high energy.
For example, the total number of electrons in Lithium is three distributed in two energy levels, closest to farthest from the nucleus- 1s2 2s1.