
Introduction | Role of the Electron Displacement in a Covalent Bond
Rarely in organic chemistry, there is a happily ever after. Things are never settled, and a chase or a pursuit is always involved.
Once the atoms tie a knot as a covalent bond (Chapters 3-Bonding in Atoms and 4-Covalent Bond), the electrons holding the atoms in place become the next major concern, so much that it determines the outcome of most organic reactions.
One begins to ponder over these electrons – are the electrons happily and evenly distributed between the two atoms? If not, then who has the major share and why? What is the impact if the electron share is uneven?
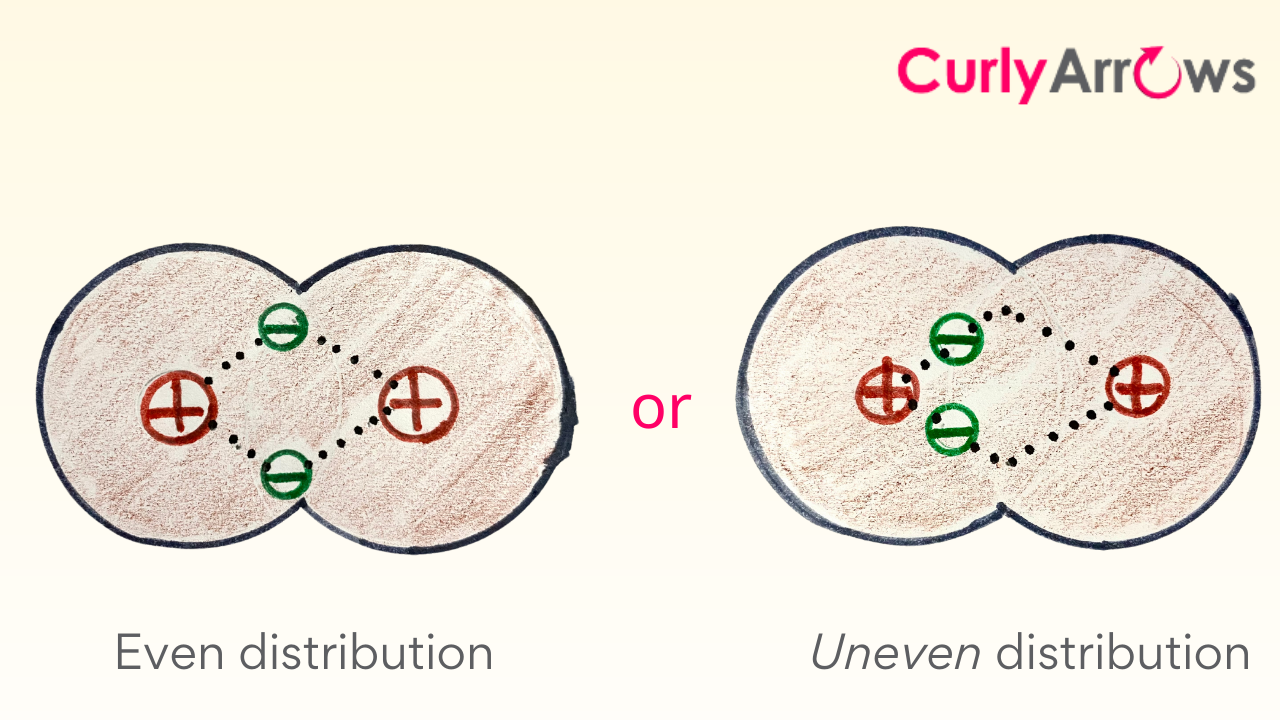
In an event where the electrons are displaced far from their usual location, beyond those two atoms in a covalent chain, we are left to face more challenging questions like how they got there, who displaced them, and whether they are happy and stable in their new location. Who are their neighbors, will they help stabilize the electrons, and so on?

So, the chapter Electronic Displacements in a Covalent Bond tries to address all these major concerns. The chapter covers in-depth all the electron-shifting effects that drive chemical reactions. The common ones are Electronegativity, Inductive, Resonance, and Hyperconjugative effects.
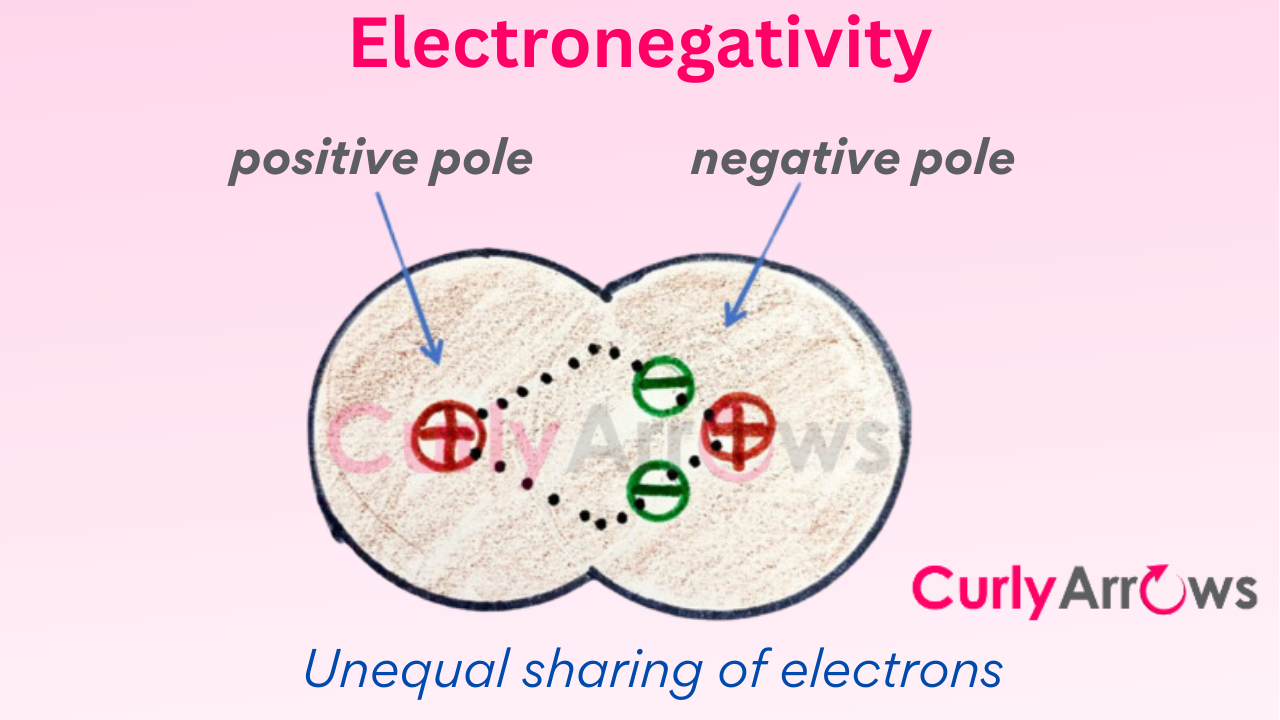
Electronegativity is when the two atoms holding the covalent bond in place do not share the bond electrons equally. Such a mismatch can create opposite poles that are a starting point for many chemical reactions. This creation of poles leads to another concept called Polarity where the strength of molecules is measured using Dipole Moment, also part of this chapter. These concepts influence molecular reactivity.
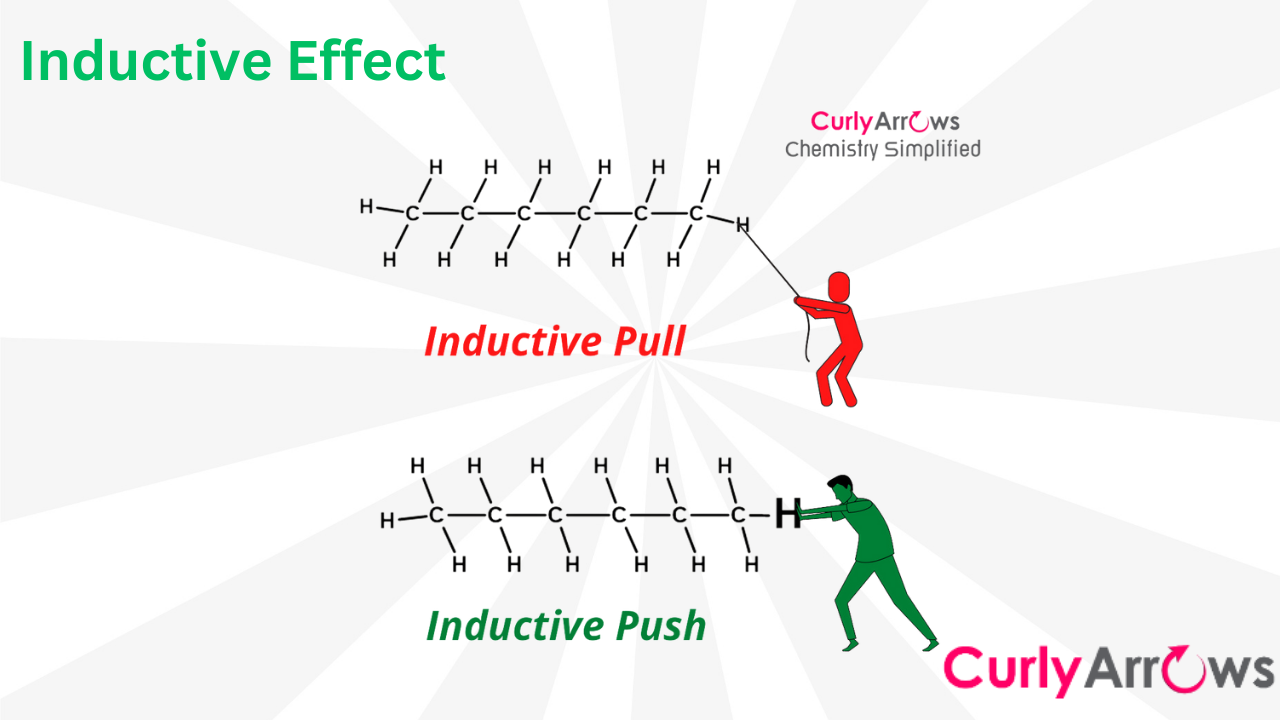
In the Inductive effect, the electron density is imagined as shifting along three or more covalent bonds. An atom or group of atoms is thought to push or pull the electron density across this distance. Such a shift can positively or negatively impact molecular reactivity.
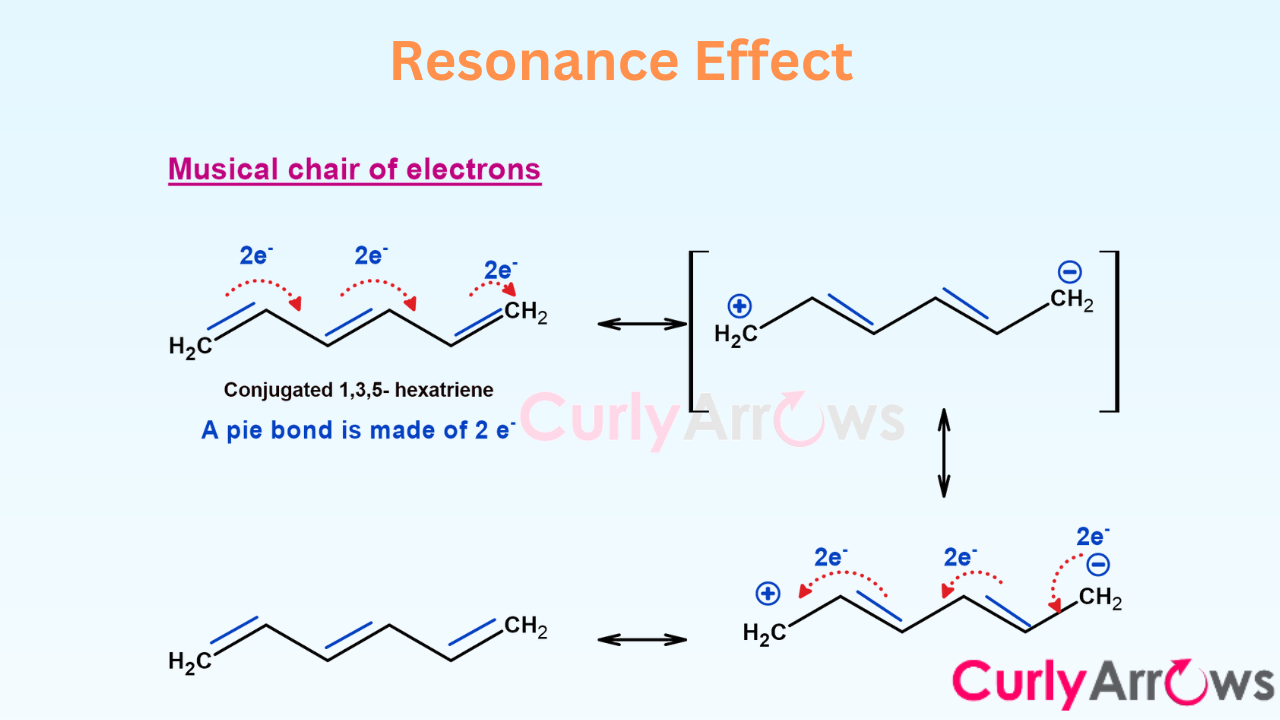
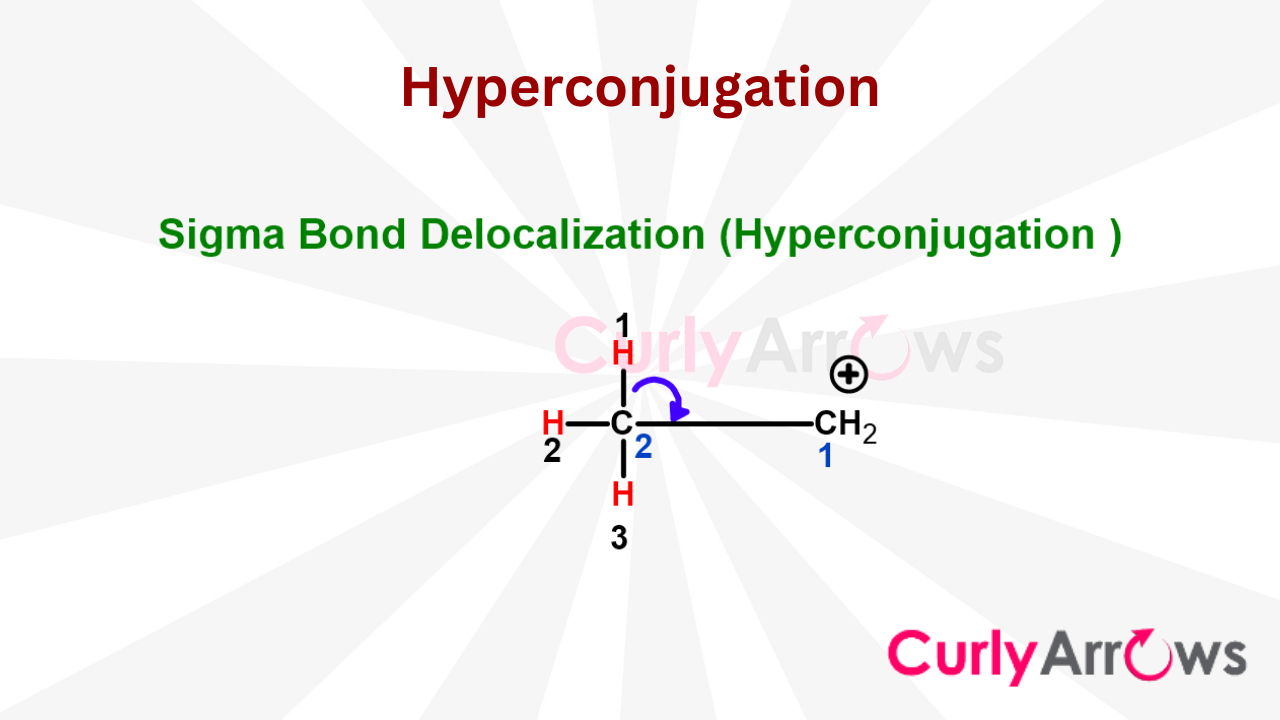
Resonance occurs when the pie electrons take off and move across the covalent bonds. It is also responsible for generating doppelgangers, aka multiple Lewis structures. When sigma electrons think of moving close by, the effect is Hyperconjugation. Both resonance and hyperconjugation effects have a huge impact on molecular stability.
We have ensured that atoms are responsible for sharing their electrons. So, we have included a chapter on Formal Charges, which is a bookkeeping method to keep atoms accountable.
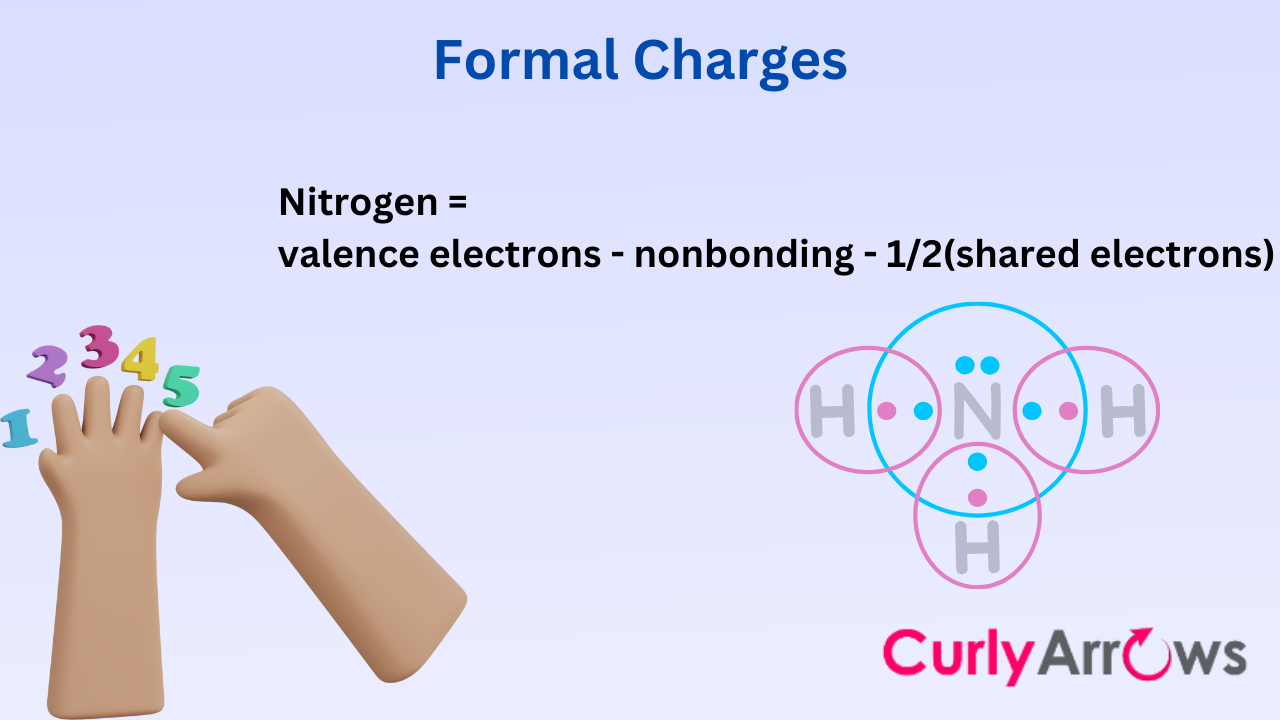
Here, we calculate every electron transaction and its net effect on the molecule, whether it develops any charge—positive, negative, or neutral.
In short, the chapter offers a bird's eye and also a zoomed-in perspective on the electron chase.

Learning chemistry requires a vivid imagination and a fun pursuit. We agree with Johann Goethe that if you wish to draw pleasure out of life, you must attach value to the world.

Our valuable contribution is understanding the importance of a covalent bond in a fun, simple way.
The following chapters provide a masterclass on all electron displacements driving organic chemistry reactions.
- Electronegativity- Introduction
- Factors Affecting Electronegativity- Atomic number, Atomic size, Shielding effect
- Factors Affecting Electronegativity-s-orbitals, Oxidation state, Group electronegativity
- Application of Electronegativity in Organic Chemistry
- Physical Properties Affected by Electronegativity
- Inductive effect- Introduction, Types, Classification, and Representation
- Factors Affecting Inductive Effect- Electronegativity
- Factors Affecting Inductive Effect- Bonding Order and Charge
- Factors Affecting Inductive Effect- Bonding Position
- Application of Inductive Effect- Acidity Enhancement and Stabilization of the counter ion due to -I effect
- Application of Inductive Effect-Basicity enhancement and stabilization of the counter ion due to +I effect
- Application of Inductive Effect-Stability of the Transition States
- Application of Inductive Effect-Elevated Physical Properties of Polar Compounds
- Is Inductive Effect same as Electronegativity?
- Resonance- Introduction and Electron Delocalization
- Partial Double Bond Character and Resonance Hybrid
- Resonance Energy
- Significance of Planarity and Conjugation in Resonance
- p-orbital Electron Delocalization in Resonance
- Sigma Electron Delocalization (Hyperconjugation)
- Significance of Hyperconjugation
- Resonance Effect and Types
- Structure Drawing Rules of Resonance (Includes Summary)
- Application of Resonance
- Covalent Bond Polarity and Dipole Moment- Introduction
- Molecular Dipole Moment
- Lone Pair in Molecular Dipole Moment
- Applications of Dipole Moment
- Formal Charges- Introduction and Basics
- How to Calculate Formal Charges (With Solved Examples)
- Difference between Formal charges and Oxidation State