Breadcrumb
- Home
- Organic Chemistry - Drawing Structures, Concepts, and Examples
The ylide is an electrically neutral molecule that has a negative carbon with an unshared electron pair (usually a carbanion) adjacent to a positive heteroatom (typically nitrogen, phosphorus, or sulfur), where both the atoms have full octets (eight electrons in their valence shell).
The bond between the carbanion and positive heteroatom is best described as a polar covalent bond with significant ionic and double-bond character, depending on the ylide type.
Learning Objective: To learn about the atomic mass number using protons and neutrons.
Skill Level – Intermediate
Prerequisites:
Learning Objective: To learn about the components of an atom and the importance of proton numbers.
Skill Level – Intermediate
Prerequisites:
Learning Objective: To learn about the size and composition of an atom.
Skill Level – Intermediate
Prerequisites:
Units of measurement in chemistry, mainly for length
| Physical Properties | Chemical Properties |
|---|---|---|
Meaning | Physical properties are those properties that can be observed or measured without changing the substances' identity; that is, there is no change in the substance's chemical structure. It does not undergo any chemical reaction that results in a structural transformation, and no new substance is formed. |
| Extensive | Intensive |
|---|---|---|
Definition | The properties that change when the size or amount of the sample changes are defined as extensive properties. That is, if the properties are dependent on the amount of matter, then doubling the quantity of matter must also double the property. |
| Qualitative Analysis | Quantitative analysis |
|---|---|---|
Aim | Identifies what compounds or functional groups are present. Therefore, the type of information obtained is descriptive. For example, whether the compound changed color, evolved gas, or formed a precipitate.
| Measures how much of a compound is present and, therefore, numerically describes its quantity. |
Dihedral Angle | Bond Angle | |
|---|---|---|
Definition |
The comparison captures the differences between valence bond theory and molecular orbital theory based on Definition, Bonding, Formation of Orbitals, Types of Orbitals Forming Bonds, Properties, Scope, Resonance Concept, and Limitations.
Aspect | Valence Bond Theory (VBT) | Molecular Orbital Theory (MOT) |
|---|---|---|
Basic Concept |
Aspect | Atomic Orbitals (AOs) | Molecular Orbitals (MOs) |
|---|---|---|
Definition | Simple regions around the nucleus of an atom where there’s a high probability of finding an electron and these regions is represented as spheres, or lobes. | A region in a molecule where electrons are likely to be found, formed by the combination of atomic orbitals of two or more atoms. |
Learning Objective: To learn about tautomerism, a subtype of structural isomerism.
Skill Level – Intermediate
Prerequisites:
Learning Objective: To learn about metamerism, a subtype of structural isomerism.
Skill Level – Intermediate
Prerequisites:
Learning Objective: To learn about functional isomerism, a subtype of structural isomerism.
Skill Level – Intermediate
Prerequisites:
Learning Objective: To learn about ring–chain isomerism, a subtype of structural isomerism.
Skill Level – Intermediate
Prerequisites:
Learning Objective: To learn about positional isomerism, a subtype of structural isomerism.
Skill Level – Intermediate
Prerequisites:
Learning Objective: To learn about chain or nuclear isomerism, a subtype of structural isomerism.
Skill Level – Intermediate
Prerequisites:
If you look at the shape of s and p orbitals before hybridization, you will notice that -
The s orbital is spherical.
The p orbital is dumbbell-shaped, where the two lobes are proportionate along one axis.
However, when these orbitals of the same atom mix to form hybrid orbitals, the newly formed shape is that of teardrops pointing in opposite directions, with one lobe more prominent than the other.
Learning Objective: To learn what is structural isomerism and its subtypes.
Skill Level – Intermediate
Chapter: Structural Isomerism
Learning Objective: To calculate the number of active hydrogen bonds per molecule and techniques for detecting hydrogen bonds.
Skill Level – Intermediate
Prerequisites:
Alpha hydrogens
Learning Objective: To learn about the impact Hydrogen bonding has on the physical properties of melting/boiling points, state, solubility, and reaction rates.
Skill Level – Intermediate
Prerequisites:
Learning Objective: To learn about different factors affecting hydrogen bond strength.
Skill Level – Intermediate
Prerequisites:
Learning Objective: To learn how an ion can amplify polarity in polar molecules.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To learn about the factors affecting ion-induced dipole interaction strength.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To learn about the ion-induced dipole interactions, their strength, and their occurrence.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To learn with examples the application of ion-dipole interactions in our daily lives.
Skill Level - Intermediate
Chapter: Intermolecular Forces
Learning Objective: To learn about the factors affecting ion-dipole interaction strength.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To study how ionic solids and dipoles of polar liquids interact and their strength.
Skill Level - Intermediate
Prerequisites:
Ions
Learning Objective: To study how molecular weight, molecular shape, functional group, and carbon number impact density.
Skill Level - Intermediate
Prerequisites:
Common names of alkanes
Learning Objective: To study which intermolecular forces and structural features affect the solubility of organic compounds in water.
Skill Level - Intermediate
Prerequisites:
Hydrophobicity
Learning Objective: To investigate which intermolecular forces are predominant in a liquid state and to study the impact of molecular weight, molecular shape, and polarity on the boiling point of liquids.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To study the impact of symmetry, carbon numbers, geometry, and polarity on the melting point of solids.
Skill Level - Intermediate
Prerequisites:
Commonly, it is accepted that a compound is an Arrhenius acid if it liberates hydrogen ions as H+ in water. In the next step, these H+ ions combine with water molecules to form hydronium ions (H3O+). The two steps can be summarised to say that Arrhenius acids are compounds that form hydronium ions, provided water is the solvent.
Learning Objective: To learn about the intermolecular forces and how they impact an organic compound's physical properties.
Skill Level - Intermediate
Prerequisites:
Structural formulas are used to display covalently bonded molecules in the order they are bonded. The atoms may bond using single, double, or triple bonds. Some structural formulas show only the atoms without displaying the complete structure with bond angles and, in some cases, even the bonds.
Atoms generally form bonding arrangements that give them filled shells of electrons like a noble gas configuration. The stability the atom aims for is that of its nearest noble gas.
For example, Lithium (Z = 3, Electronic Configuration = 1s2, 2s1) of the second row would prefer losing one electron to become Li+ (Z = 3, Electronic Configuration = 1s2) having an electronic arrangement similar to its nearest noble gas Helium (Z = 2, Electronic Configuration = 1s2).
Learning Objective: To learn about a few common reactions that carbon-centered free radicals undergo in organic chemistry.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To learn the two different ways a free radical reacts and the mechanism it undergoes.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To learn how to predict the stability of a hydrocarbon to form a free radical using the dissociation energy value of the C-H bond.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To study the structural features that contribute to the stability of a carbon-centered free radical in organic chemistry.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To study the structural features of a carbon-centered free radical in organic chemistry.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To learn about different ways by which the formation of a carbon-centred free radical occurs.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To learn about various methods by which a carbanion reacts with other positive centers in organic chemical reactions.
Skill Level - Advanced
Prerequisites:
Learning Objective: To compare different molecular structures with their pKa values to understand which structures can easily form and effectively stabilize a carbanion.
Skill Level - Advanced
Prerequisites:
Acidity
Learning Objective: To learn different methods to stabilize a carbanion.
Skill Level - Advanced
Prerequisites:
Learning Objective: To learn about the three ways carbanion intermediates are generated.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To learn about the structure, type, and nature of the carbanion intermediate part of organic chemistry reactions.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To understand the four common organic reactions that generate carbocation intermediates.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To study the fate of a carbocation intermediate in a chemical reaction.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To learn about the structural feature that stabilizes also destabilizes the carbocation intermediate.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To study the two methods of how carbocation forms.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To study carbocation as the reactive intermediate - formation, structure, and types.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To study what reactive intermediates are, their types, and their role in organic chemistry.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To learn through an example how a reaction outcome depends on the conformation of the reactant.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To study the role of solvents in conformational isomerism.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To understand the conformations molecules containing lone pair(s) mostly adopt.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To study the importance of conformational analysis in organic chemistry.
Skill Level - Intermediate
Chapter: Stereochemistry
Sub-topic: Conformational Isomerism
Learning Objective: To study the various stabilizing and destabilizing interactions affecting the stability of conformations.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To study the various terminologies for single-bond and double-bond-containing molecular conformations.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To study the common names of open-chain and closed-chain conformations and their stability.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To learn the various methods used to draw conformational isomers.
Skill Level - Beginner
Prerequisite:
Chapter: Stereochemistry
Learning Objective: To get familiar with conformational isomerism, what it means, how it occurs, and also differs from other forms of isomerism.
Skill Level - Beginner
Prerequisites:
Learning Objective: To study the ambident nucleophiles and how and from which end they attack the substrates to undergo chemical reactions.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To study the three types of addition reactions that nucleophiles undergo.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To study the three broad types of displacement reactions that nucleophiles undergo.
Prerequisites:
Learning Objective: To learn about the trend and order of nucleophilicity for the p-block elements in the periodic table
Prerequisites:
Electronegativity trend of the elements from the Periodic table
Learning Objective: To study the nucleophiles that function as sigma bond donors that form Carbon, Hydrogen, and Halogen bonds with the substrate.
Prerequisites:
Electronegativity trend of the elements from the Periodic table
Learning Objective: To study the behavior of pi bonds containing nucleophiles seen in organic chemistry reactions.
Prerequisites:
Learning Objective: To study the types of nucleophiles commonly encountered in organic chemistry and their behavior in a chemical reaction.
Prerequisites:
Learning Objective: To study the formation and different categories of electrophiles commonly seen in organic chemistry.
Prerequisites:
Learning Objective: To study the various examples of organic reactions where the leaving group departs from a molecule and also understand its various types.
Prerequisites:
Displacement and Elimination type of reaction (Chapter- Common Types of Reactions)
Learning Objective: To compare various groups on their ability to break off or 'leave' from a molecule and, in that process, classify them as good or bad leaving groups.
Prerequisite:
Learning Objective: To determine the factors responsible for the stability of a leaving group. These factors decide whether the leaving group is good or bad.
Learning Objective: To study lone pair's role in chemical reactions as electron donors.
Prerequisites:
Learning Objective: To study the impact of lone pair on a molecule's polarity and dipole moment.
Prerequisites:
Learning Objective: To study how the presence of a lone pair on the central atom of a molecule contributes to Hydrogen bonding, a type of intermolecular attractive force.
Prerequisites:
Learning Objective: To investigate how the presence of a lone pair in a molecule affects its bond angle and, therefore, affects its shape.
Prerequisite:
A dihedral angle is obtained when two planes pass through three atoms and two bonds, of which one bond is common to both planes.
In the below example, plane 1 passes through X-C-C, containing three atoms and two bonds. Similarly, the plane 2 passes through C-C-Y. The separation between the two planes or the angle of intersection is denoted using a dihedral angle (ϴ) in degrees (o).
| Alkanes | Alkenes | Alkynes |
|---|---|---|---|
Bond type | Class of organic compounds made of only carbon and hydrogen atoms (hydrocarbon), linked using a single covalent bond.
| Class of hydrocarbon compounds with at least one carbon-carbon double covalent bond. | Class of hydrocarbons containing carbon atoms held in a triple covalent bond. |
| Atomic Number | Mass Number |
|---|---|---|
Represents | The total number of protons present in the nucleus of an atom. | Total count of protons and neutrons present in the nucleus of an atom. |
Symbol | Z (AZE) Found at the subscript of the AZE notation while representing the element (E). |
Organic Compounds | Inorganic Compounds | |
|---|---|---|
Based on source (Older definition) | Compounds isolated directly or indirectly from living organisms such as plants and animals were earlier called Organic compounds (based on the Vital Force Theory). |
Organic chemistry is a branch of chemistry that studies transformations at three levels- macroscopic (visual changes like rotting of an apple), microscopic (atoms, elements, electrons, involved), and symbolic (representing changes using bond breaking and making, electron transfer arrows). It, therefore, studies structures, composition, properties, formation, and reactions of various compounds, mainly carbon-containing compounds, to fully understand transformations.
The discovery of isomerism marked the advent of structural chemistry, where it became significantly important to establish structural formulas, welcome the abundance of structurally diverse compounds, classify them, and study their individual properties and impacts.
Around the early 1800s, scientists began to notice the presence of isomerism in all types of compounds; however, at the time, it was referred to using different names.
The term allotrope was used to describe different varieties of an element that existed in different physical forms, such as diamond or graphite for carbon or monoclinic or rhombic for sulfur. Polymorphism was used to explain compounds that occurred in more than one crystalline form, like aragonite and calcite, both crystalline forms of calcium carbonate.
Accidental discoveries and insatiable curiosity fuel chemistry.
One such significant discovery is of isomerism, which also occurred around the period when the faith in vitalism, a belief that organic molecules must only come from living organisms, was dwindling.
It was a pleasant surprise to learn that urea, a by-product of urine produced by human kidneys, could also be synthesized by heating ammonium cyanate, an inorganic compound.
Solution
The condensed structural formula disguises the molecule's true vastness by revealing only the number of atoms present. What remains hidden from sight are the bond connections, the bond angles, and their three-dimensional arrangements, so what we observe is just the molecule's literal condensed form.
Nucleophiles are electron-rich species that can donate a pair of electrons and form a new covalent bond with an electron-deficient counterpart called the electrophiles. That is why electrophiles are referred to as lovers of electrons.
The nucleophiles donate electrons as lone pairs, pie bonds, or sigma bonds.
Read- What are nucleophiles?
While nucleophiles donate two electrons in exchange for forming two-electron covalent bonds with the electrophiles, the negative charge due to the two electrons is not obvious in many instances. Neutral molecules like Benzene, water, and ammonia can act as nucleophiles.
A broad way to categorize nucleophiles is as charged or neutral (uncharged).
Following the footsteps of Gilbert N. Lewis in understanding the nature of the covalent bond, Linus N. Pauling stumbled upon an interesting observation. Pauling noticed that the nature of the chemical bond could be explained using a scale or continuum.
The bond angle is the angle at which two adjacent bonds converge and meet at the central atom in molecules.

The very premise of a covalent bond is electron sharing. As two atoms share one electron each to form one covalent bond, they may likely share more than once and form more bonds. This information on the number of connections between two atoms is revealed from the Bond Order.
So, the Bond Order measures the number of bonds between the two atoms in a molecule. The number can be integers like 1, 2, or 3 for single, double, or triple bonds or non-integers like 0.5, 1.3, 1.5, etc.
An important thing about an atom is that it is made of three main components- protons, neutrons, and electrons.
August Kekulé (1866) was the first to propose the alternating single and double bond structure of benzene that can only be possible if carbon could join other carbon atoms and form a ring.
The first time the use of two dots or a colon (:) to represent two electrons acting as connecting links between the two atoms was proposed by G.N. Lewis in 1916.
In his landmark paper, ‘the atom and the molecules,’ he attempted to draw molecular structures using dots to represent the covalent nature of the chemical bonds, now popularly known as the Lewis structures.
So, a molecule of Cl2 was Cl : Cl, two electrons connecting two Cl atoms.
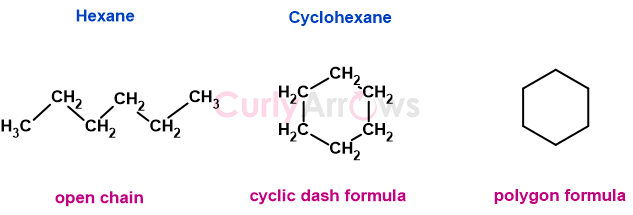
Organic compounds in which carbon atoms are joined in a ring, unlike the open-chain counterparts, are represented using the polygon formula. These cyclic polygons do not show carbon and hydrogen atoms. The corner of a polygon represents a carbon atom with its required number of hydrogen atom(s), and the sides of the polygon represent a carbon-carbon bond.
Molecular models are tools used for classroom or research to visualize better and interpret three-dimensional structures of organic molecules. The toolkit can be made of wood, plastic, metal, or other materials.
These models are digitally represented using computer graphics covered under molecular modelling, a branch of chemistry that uses computational and theoretical methods to model and mimic the molecule's behaviour in chemical and biological environments.
Carbon and Hydrogen containing bonds are chemically inert; still, they form the backbone of many organic compounds. They provide a skeletal framework that can contain other heteroatoms or functional groups.
Such a carbon-hydrogen-containing chain of compounds together with other atoms or groups of atoms are conveniently represented using zig-zag lines, called the bond-line or skeletal formula.
Certain conventions are used to represent three-dimensional structures of organic molecules on paper. Using simple lines, solid (dark), and dashed wedges, a 3D image of a molecule can be perceived on two-dimensional paper.

The condensed structural formula provides the shortest way to understand the atoms and their numbers, their connection with respect to each other, and therefore, visualize the compounds' structure without elaborately drawing it, but by writing it in a single line.
So, if the elaborate structure of butanoic acid look like A, the condensed structural formula would look like B.
Learning Objective: To study the octet rule by G.N. Lewis, considering both ionic and covalent bonding, with examples.
Skill Level – Intermediate
Prerequisites:
a) Carbon and Silicon
b) Nitrogen and Phosphorus
c) Oxygen and sulfur
d) Magnesium and Calcium
In his landmark paper, 'The Atom and the Molecule,' G.N. Lewis attempted to describe linkages between the atoms to understand the nature of covalent bonds.
He used dots to represent an atom’s valence electrons and argued that the atoms share their valence electrons to form one, two, or three bonds until they attain a stable octet electron configuration. An exception is the Hydrogen atom that attains a duplet configuration.
Learning Objective: To study the limitations of the octet rule, with examples.
Skill Level – Intermediate
Prerequisites:
d-orbitals
Carbon is naturally inclined to form new bonds, and in this quest, it may break away from the old ones. The atom or group of atoms that leaves the carbon chain to make way for the new bond is called the leaving group(s).
Such a bond dissociation can happen from a saturated, unsaturated, or carbonyl carbon.
The longest carbon chain is the parent chain containing the principal functional group.
The principle functional group is always given the highest priority, lowest number (1 before 2, 3, 4...), and assigned as a suffix while writing the nomenclature. Therefore, the numbering of the parent chain always starts from the principal functional group.
IUPAC, an international body engaged in developing standards for naming chemical elements and compounds, has laid several rules known as 'IUPAC rules' to identify a molecule by its parent alkane chain and the functional group. It has assigned various suffixes to identify organic chemistry's commonly seen functional groups.
Organic compounds predominantly consist of Carbon and Hydrogen atoms, also known as hydrocarbons. However, most organic compounds also contain other heteroatoms like O, N, S, and halogens as the functional group.
Out of the many ways to classify a structure, the functional group classification based on the attached atoms is listed below.
Learning Objective: To study with examples the hydrogen bond donors and acceptors.
Skill Level – Intermediate
Prerequisites:
Radical initiators are chemical species that quickly produce free radicals by light and heat exposure. Examples include Halogens (Cl2), AIBN (R-N=N-R), peroxides (R-O-O-R), acetophenone, benzophenone, etc.
A radical reaction usually happens in three steps- Initiation (creation of free radicals), Propagation (multiplication and growth of radical reaction), and Termination (stopping of the free radical reaction).
Radical inhibitors are chemical species that inhibit radical reactions by halting the chain propagation step.
Examples of such inhibitors are Hydroquinone, BHT (2,6-di-t-butyl-4-methylphenol), etc.
Free radicals are generated due to the homolytic bond cleavage, a type of bond breaking where each atom holding the two-electron covalent bond gets one electron. Such a homolytic bond cleavage requires an input of energy, either in the form of heat or light.
Inorganic chemistry describes oxidation in two ways. The first method is through the loss of electrons, and the second is by the increase in oxidation number.
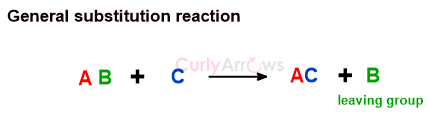
In substitution reactions, an atom or group of atoms is displaced and replaced by another atom or group of atoms. The displaced atom is called the leaving group. The rest of the molecule remains unaffected.
Elimination reaction involves the loss of two groups and the formation of a new bond.
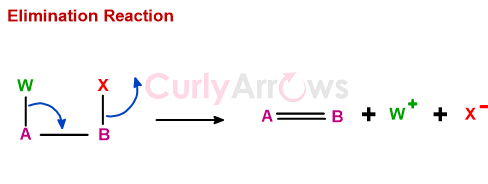
Elimination reaction takes place either by heterolytic or pericyclic mechanism.
A decomposition reaction breaks down the substrate into two or more smaller, simpler molecules.
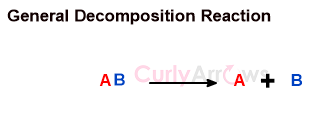
The decomposition reaction is initiated by obtaining energy from heat, light, or electricity sources.
Depending on the source, the decomposition reaction is of three types-
An atom or group of atoms migrate from one position to another; such reactions are rearrangement reactions.
Post rearrangement, the molecule formed has the atoms arranged differently but has the same molecular formula as the parent. Therefore, such rearrangements create structural isomers, compounds that differ in structure but have the same molecular formula.
Related Reading - Structural Isomerism
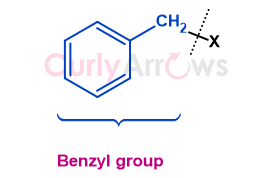
A benzyl, abbreviated as Bn, refers to the phenylmethyl group (C6H5-CH2-), consisting of a benzene ring attached to a methylene (-CH2-) group.
Learning Objective: To study the history, nature, examples, and types of free radicals, a reactive intermediate in organic chemistry.
Skill Level - Intermediate
Prerequisites:
A carbocation is a positively charged, electron-deficient ion of carbon with only 6 valence electrons from the preferred 8 for covalent bonding. Therefore, it acts as an electron-accepting reactive intermediate in many organic reactions.
After accepting two electrons, it can transform from a less stable to a more stable uncharged molecule.
A carbocation is a positively charged, electron-deficient carbon ion that acts as a reactive intermediate in many organic reactions.
The central carbon in a carbocation has only 6 valence electrons instead of the preferred 8 by covalent bonding. As a result, it has only three covalent bonds and, therefore, trivalent.
A carbocation is a reactive intermediate with only 6 valence electrons from the usual 8 electrons for carbon by covalent bonding. This makes the carbon atom bear a positive charge and possess three bonds instead of four.
A carbocation is a positively charged, electron-deficient carbon atom that acts as a reactive intermediate in many organic reactions.

In ionic compounds, a metal atom loses an electron, forms a cation, and another nonmetal atom gains it, creating an anion. Thus, there is a complete transfer of electrons between the two atoms. The ions are part of a fixed crystal lattice and immobile in the solid state. For example, NaCl, KBr.
Metallic solid consists of several metal atoms of the same kind bonded together closely. Due to their large size, metals easily lose their outermost valence electrons. The delocalized valence electron drifts and is now available for the other nuclei of metal atoms. Several delocalized electrons are comparable to a sea of electrons.
Several characteristic properties of the metals are due to the nature of the metallic bond. Some unique properties include-
1. High Melting and Boiling points:
A metallic bond is a chemical bond seen in metals consisting of tightly bound metal atoms of the same type.
Metals are large atoms that do not firmly hold their outermost valence electrons and easily lose them. Once the electrons are lost, the metal atoms become positively charged, called kernels. The position of these kernels is fixed to avoid repulsions and is part of the solid structure.
Van der Waal’s interactions are the weak intermolecular forces of interactions. The Van der Waal’s forces are comprised of three forces. Their different names and the order of their strength is-
Dipole-Dipole > Dipole-induced dipole > Induced Dipole-Induced Dipole
Debye intermolecular forces of attraction occur between polar and nonpolar atoms or molecules.
When a polar molecule is in the vicinity of a nonpolar molecule, the electronegative end of that polar molecule attracts the electron cloud of the adjacent nonpolar molecule, thereby distorting it and inducing a pole separation. So, the polar molecule is said to have induced polarity in the neighboring nonpolar molecule.
Debye intermolecular forces of attraction occur between polar and nonpolar atoms or molecules. When closely placed with a polar molecule (electronegativity difference between 0.5-1.7), the electron cloud of the nonpolar atom (or molecule) is influenced and distorted, leading to the formation of temporary dipoles. As the polarity is induced (forced) in the otherwise nonpolar atom, the Debye forces are said to show an Induction effect.
If the size of a nonpolar atom is greater, it means that the atom has a higher number of electron containing orbitals. The outer electrons are away from experiencing the inward attractive pull of the nucleus (nuclear charge) and capable of undergoing electron-cloud distortion or polarization.
Debye interactions require polar molecules to show polar-nonpolar type of intermolecular attractive interactions. Therefore, the polar atoms participating in Debye interactions must have comparatively high electronegativity to show stronger interactions. If the electronegativity difference (0.5-1.7) is greater in a polar molecule, the polarity it induces on the neighboring non-polar atom will also be more significant, leading to stronger Debye interactions.
The strength of polar-nonpolar interactions is affected by the atomic size of the nonpolar atom (or molecule) and the electronegativity of the polar molecule.
A polar molecule has natural poles. Therefore, when next to a nonpolar molecule, it will try to magnetize it temporarily.
The negative end of the polar molecule will repel the nonpolar molecules’ moving electron cloud, or the positive end will attract its electrons, which can contribute to significant distortions. Such a forced activity can induce a temporary pole separation in the nonpolar molecule.
London dispersion forces are weak intermolecular forces found in all atoms and molecules. However, it is an exclusive force binding the nonpolar molecules due to their lack of functional groups, avoiding competing interactions.
London Dispersion forces are seen in all the atoms and the molecules; however, it is the exclusive force binding nonpolar molecules. Nonpolar molecules are made of two or more atoms, and the electronegativity between two atoms in a covalent bond is less than 0.5. Therefore, no charges are separated to create poles in nonpolar molecules or atoms. Despite not having poles, nonpolar molecules interact using a weak force of attraction known as the London Forces.
All atoms and molecules show London Dispersion Force, a primary way atoms and molecules interact. Most of the time, molecules have additional stronger forces than the London Dispersion Force, which is the weakest. Therefore, London Dispersion forces are exclusively seen in the nonpolar molecules since they don't have other competing functional groups leading to other interfering intermolecular interactions.
A nonpolar molecule is made of two or more atoms where the electronegativity difference between the atoms is less than 0.5. Therefore, the electron density in the bond is equally distributed, and there is no concentration of electrons on one atom to create a charge separation rendering the molecule essentially without poles or nonpolar. Such nonpolar molecules like H2, CH4, He, etc., will interact using the London dispersion forces without forming any positive or negative pole.
London dispersion force is the weakest of the three van Der Waal forces of interactions universally found in all the atoms and molecules. It is caused by the constant fluctuations in the electron position, creating a temporary dipole. When other nonpolar molecules are in its vicinity, the instantaneous temporary dipole formed earlier distorts the electron cloud of an adjacent molecule, creating another dipole.
London dispersion forces are the weakest intermolecular forces among all the van der Waal forces of interactions. This interaction occurs in molecules due to electrons' continuous random motion, creating a temporary dipole. The instantaneous dipole in one molecule induces a dipole in the others, causing dispersion. The molecules then start to show intermolecular attraction.
Hydrogen bonding is a type of electrostatic interaction occurring in molecules in which the H atom is sandwiched between highly electronegative atoms like F, O, and N, one to which it is covalently bonded. For example, HF, H2O, NH3, etc.
Keesom forces occur in polar molecules where the atoms have a considerable electronegativity difference, resulting in the charge separation and formation of positive and negative poles. The negative pole of one molecule electrostatically interacts with the positive pole of the neighboring molecule. Consequently, all the polar molecules in the medium try to orient their dipoles to be in alignment for such electrostatic interactions.
Polar molecules show Keesom forces. Polar molecules have heteroatoms that differ in electronegativity values such that the electronegativity difference between the two atoms in a polar covalent bond is greater than 0.5 but less than 1.7. For example, HF, HCl, R-OH, etc.
A molecule is said to be polar if the bond electrons between the atoms in a covalent bond are unequally distributed, creating two ends. The electron redistribution is marked by showing the separation of charges as a partial charge above the atom's symbol. Therefore, a polar molecule always has a positive and a negative end resulting in a dipole. The pulling of bond electrons by an atom is attributed to its electronegativity.
There are three types of Van der Waal’s forces: Keesom Forces, Debye forces, and London Dispersion forces.
The van der Waal forces of interactions are weak intermolecular forces. For the atoms or molecules show these interactions only under certain conditions -
1) Distance- van der Waal forces mainly depend on the distance between the associated molecules. These forces become very weak if the distance between the molecules or atoms is greater. These forces are relatively stronger if the atoms or molecules are grouped closely.
There are three types of Van der Waal’s force- Keesom Forces, Debye forces, and London Dispersion forces. Amongst the three types, the London Dispersion forces are the weakest interactions.
Van der Waals forces are weak intermolecular attractive forces that occur in polar and nonpolar atoms or molecules due to the shift in their electron positions.
The electrons shift to form electron-dense and electron-deficient poles. Some poles are permanent due to the nature of the atom in a molecule, while others are induced poles.
Van Der Waals forces are weak intermolecular attractive forces that hold closely various atoms and molecules without any physical linkage or bond.
The atoms or molecules participating in van Der Waals interactions are neutral, polar, or /and non-polar ones. Ion interactions are stronger and are classified separately from the weaker van Der Waal interactions.
Solution
Solution:
Learning Objective: To learn about the average atomic mass and how to calculate it from its isotopic mass and natural abundance.
Skill Level – Intermediate
Prerequisites:
Dalton, in 1804, in his work on ‘Atomic Theory,’ proposed the law of chemical combination to explain how atoms form compounds. According to him, atoms of different elements combine in a simple whole-number ratio to give compounds.
For example, two Hydrogen and one Oxygen combine to form water (H2O), or four Hydrogens and one Carbon combine to form methane (CH4), or one Nitrogen combines with three Hydrogens to give ammonia (NH3).
Learning Objective: To study Vander Waals Debye force, interactions, and the factors affecting their strength.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To study Vander Waals Keesom's force, interactions, and strength.
Skill Level – Intermediate
Prerequisites:
Learning Objective: Study Vander Waals London forces, interactions, and strength.
Skill Level – Intermediate
Prerequisites:
The atoms that form covalent bonds are the nonmetals (p-block elements), and they must have an electronegativity difference lower than 1.7. These values also help to classify the covalent bond into two kinds, polar and nonpolar.
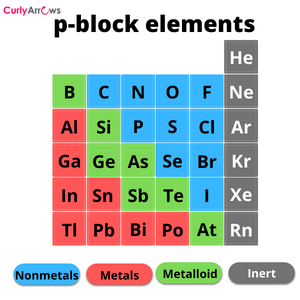
Once each electron of the two atoms forms the covalent bond, the electrons are simultaneously distributed between them so that they belong to both the atom’s nuclei. This area between the atoms is called the region of electron density.
Once the outermost valence electrons of the atoms are engaged in the covalent bond formation to make a molecule, some electrons withhold themselves from the bond-making process. This divides the electrons in the molecule into two parts- bond pair and lone pair.
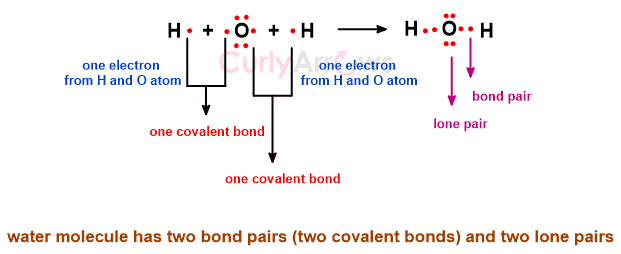
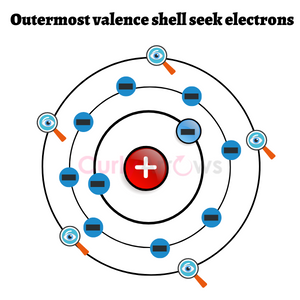
The atoms that embark on the journey of bond formation are likely to have an electron imbalance. They are less stable and become highly reactive. These atoms seek 8 electrons in their outermost shell to lower their reactivity and achieve stability.
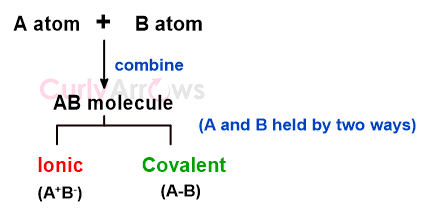
The Covalent bond is one of the two linkages by which atoms join to form molecules and is therefore classified under chemical bonds.
The atoms or group of atoms are classified based on the Inductive effect as electron-withdrawing (-I) or electron-donating (+I) relative to Hydrogen.
The common functional groups showing +I and -I effect are:
+I group | -I group |
|---|---|
O- |
Inductive, along with resonance effects, are permanent effects.
The atoms or groups of atoms causing the inductive effect are part of the molecule. Depending on the atom's nature (electron-withdrawing or donating), the groups causing the molecule's inductive effect can impact the molecule's stability and physical and chemical properties.

The Inductive effect is the outcome of Electronegativity; therefore, they are different and not the same.
Pre-Requisite Reading: Lewis Structures, Types of reactions, Using curly arrows for electron movement, Identifying Functional groups.

Pre-Requisite Reading: Resonance
Branched alkyl chlorides (20 or 3o) cannot be used as a reagent in FC acylation reaction. The branched alkyl chlorides on forming acylium ion undergo the loss of CO to create a stable alkyl cation. The 20 or 3o alkyl cation reacts with benzene to form alkyl benzene.
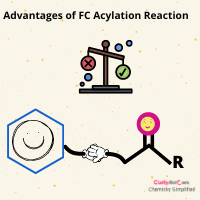
The drawbacks of Friedel Craft Alkylation, such as polyalkylation and rearrangements, are overcome by FC Acylation Reaction.
The stability order of different alkene substitution patterns is measured by comparing their heat of hydrogenations (denoted as ∆Ho) in kJ/mole or kcal/mol.
Alkene is a double bond containing hydrocarbon compound. Since the hydrocarbon compounds can be short or long chained, the double bond's position could be anywhere along the hydrocarbon chain.
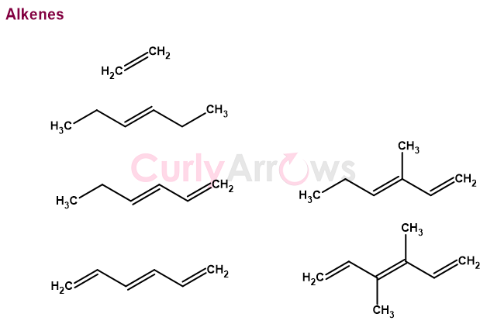
Alkenes are double bonds containing molecules. These double bonds are called pie bonds and are made up of two electrons. Due to the presence of the pie electron cloud, alkenes are electron-rich species and interact with electron-deficient species, the electrophiles. The reaction between alkenes and the electrophiles is an addition reaction.
Sharing resources is essential to build a harmonious world. When the resources are shared ineffectively conflicts emerges. A similar principle extends to Chemistry.
Atoms are in a state of harmony when the neighbouring atoms shares electrons. When the sharing stops, Chemical reactions triggers.
The imbalance in sharing electrons influences the molecule’s polarity, reactivity, and physical properties. The disagreement on sharing is brought about by electronegativity.
Electrophiles love electrons, and without them, they become unstable. And to reach a stable state, the electrophiles accept electrons from electron-rich nucleophiles.
Therefore, the electrophiles are electron-deficient species and acceptors of electrons.
Pre-requisite: Nuclear charge, Valence electrons
Stovetop cooking routinely requires cast iron or aluminium pans but never a glass pan or a pan made of silicon. That’s because the glass pan will shatter at high temperatures, and the silicon pan will burn away.
Water at room temperature is liquid in nature due to the hydrogen bonds holding the water molecules (H2O). The close association is hard to break; therefore, water boils at a high temperature of 100oC. The high temperature breaks the Hydrogen bond links to free individual H2O molecules.
A covalent bond holding two atoms is made of two electrons. The bond can cleave or break in two ways - equally (homolytic fission) or unequally (heterolytic fission).
A heterolytic bond cleavage results in unequal bond-breaking where one atom in the bond retains both the bond electrons.
A chemical bond strength is a force holding the atoms in a bond, and separating such atoms requires energy input. The bonds are of two types- Intermolecular and Intramolecular bonds.
Intramolecular bonds join the atoms in a molecule, whereas Intermolecular bonds are only responsible for closely associating the molecules. Therefore, Intramolecular bonds require higher energy to break than intermolecular bonds.
Organic chemistry and human beings both pursue betterment and advancement.
Humans engage in welfare activities to improve the well-being of others, tackle inequality, and create strong bonds. The same principle is also observed in organic chemistry.
Lone pair is a set of electrons present in an atom’s valence shell that did not participate in any bond-formation reaction. Since they refuse to bond with the other atoms, they are also called the non-bonding electrons. While drawing the molecules’ structure, the lone pair electrons on shown as dots (..) above the atom.
Russian chemist Vladimir Markovnikov proposed Markovnikov’s rule in 1869 to predict the regiochemistry of addition reactions between unsymmetrical alkenes/ alkynes and hydrogen halides to form alkyl halides.
According to the rule, ‘An alkyl halide is formed in a reaction between an unsymmetrical alkene or alkyne and hydrogen halide (HX) when the negative part of the reagent (X-) attaches to the alkene carbon that has fewer number of hydrogen atoms across the double bond.’
Learning Objective: To study what Van Der Waal forces are in chemistry and their example.
Skill Level - Intermediate
Prerequisites:
Prerequisite Reading: Structural Isomerism, Types of Hydrogen, Free radical halogenation
The Hydrogens attached to a Carbon atom are said to be equivalent if they are in a same chemical environment. Same chemical environment means that under a reaction condition, these hydrogens would lose their identities of being attached to separate Carbons and behave like an identical set.
The DBE calculation uses the general molecular formula to find the presence of unsaturation in a compound. The unsaturation is calculated in levels or degrees.
The lowest degree of unsaturation (DOU) indicates minimum unsaturation, where there is the least loss of hydrogens to form a pie bond or a cyclic ring, like in cycloalkanes.
General Molecular Formula
A halogen derivative of an alkane is called a Haloalkane, where the hydrogen is replaced with a halogen.

The common or the trivial name is Alkyl Halide.
A fun mnemonic to remember a few common preparation methods of haloalkane is hidden in the common name.
The three common hybridization states are - sp3, sp2, and sp. The sp3 (pronunciation: ess-pee-three) hybridization of Carbon explains its four bonds’ tetravalency, shape, and equivalency.
A Haloalkane is the Halogen derivative of an alkane obtained by replacing of one or more hydrogen atoms.
For example, when one Hydrogen of an alkane, Methane, is replaced with a Halogen, for instance, Chlorine, the haloalkane obtained is chloromethane.
Alkane Haloalkane
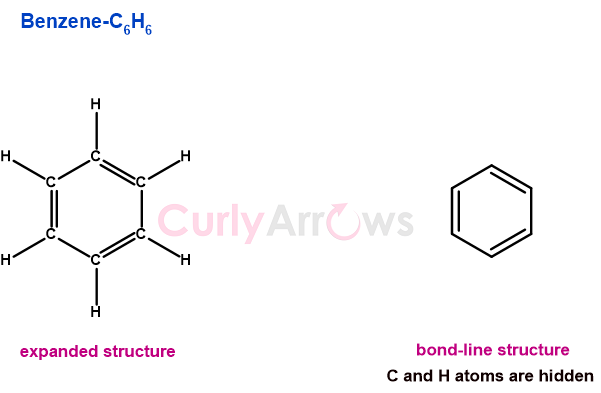
It is easy to identify the simplest aromatic hydrocarbon Benzene that is made of only two atoms- Carbon and hydrogen. Benzene is a planar, cyclic ring molecule with six sides. It has an alternating double bond that contributes to its extra stability by endowing it with resonance, a phenomenon of electron delocalization. The molecular formula of Benzene is C6H6 and the structure is-
The nomenclature is mainly used for sp3 hybridized Carbon of an alkane. The Carbon of alkyl halides, alcohols, carbocations, and the Nitrogen of amides and amines are classified by this method.
The replacement of the hydrogen in acetic acid (H-CH2-COOH) with the chloro gives chloroacetic acid (Cl-CH2-COOH). Therefore, Acetic acid and Chloroacetic acid have carboxylic acid (R-COOH) as the principal functional group.
Lattice points are the positions an atom, ion, or molecule can occupy in a crystal giving it its shape and characteristics. If any of these particles go missing or are not in a correctly ordered arrangement, it can introduce a defect in the crystal. Lattice points are essentially a crystalline framework.
1) The first step is find out the Degree of Unsaturation (the number of double bonds or the presence of a ring) in the compound.
The formula is,

Alkenes undergo addition reactions where it loses one pie bond to form two new sigma bonds to add a reagent across the double bond.
The Lewis dot structures are used to show the shared electron pairs between the bonded atoms in the molecule and the lone pairs of electrons if any. The representative structures follow the octet rule wherein the atoms combine by either transfer of electrons (loss or gain) or by sharing of valence electrons in a way that the valence shell of the atoms attains the octet configuration.
The electrons involved are shown as dots. A single bond is made up of two electrons, a double four and a triple bond six. Similarly, an atom can have one, two or three lone pairs of electrons.
The electronic configuration of two elements A and B are -
A- 1s2 2s2 2p6 3s2 3p6 4s2
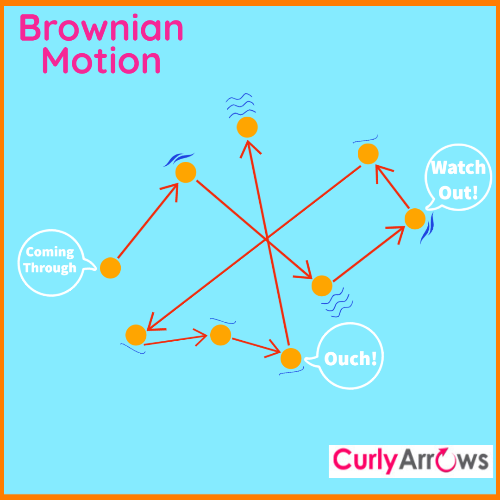
On a fine morning in 1827, botanist Robert Brown had no idea that a simple observation through his microscope would lead to the discovery of invisible matter.
In the presence of moisture, PCl3 undergoes hydrolysis to release fumes of HCl gas.
PCl3 + 3H2O → H3PO3 + 3HCl
The reaction mechanism is shown as,

The oxidation number of Phosphorus (P) is the unknown here. Let us consider it as x.
The Oxidation Number of Hydrogen (H) is + 1
The dipole moment, a product of charge difference (q) and the distance (d) between the centres of positive and negative charges (µ = q x d), is also directional. The direction is indicated by an arrowhead, which points towards the most electronegative atom, representing the direction of the dipole moment.
Understanding the failure of Benzene to undergo an Iodination reaction requires insight into multiple aspects of the reaction mechanism and the nature of the reactants. Here in the article, we will explore a few methods of increasing the reactivity of Iodine using different reaction conditions.
In the carbon-halogen covalent bond, the halogen due to its higher electronegativity pulls the electrons in the bond towards itself. The unequal sharing of the bond electrons results in carbon to be electron-deficient and halogen, electron-rich. This charge redistribution is shown with a partial positive charge (δ+) on the carbon and a partial negative charge (δ-) on the halogen.
Friedel Craft acylation reaction is a type of electrophilic aromatic substitution reaction wherein the hydrogen of the aromatic ring is substituted with the acyl group (R-CO-), and a new Carbon-Carbon bond is formed. Such an acylation reaction requires a Lewis acid catalyst such as AlCl3 and an acid chloride (R-CO-Cl). The byproduct formed is HCl.
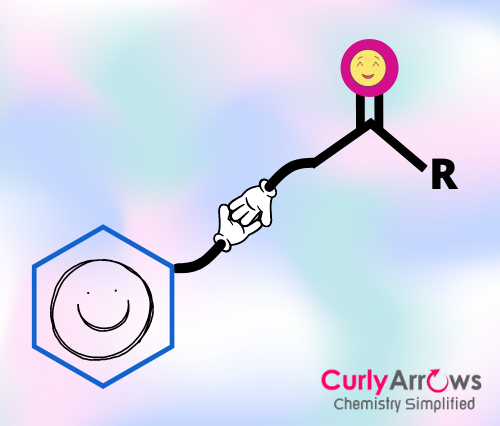
Friedel Craft Acylation reaction is an electrophilic aromatic substitution reaction involving acylium ion as the electrophile.