Introduction to Atoms: The Building Blocks of Matter
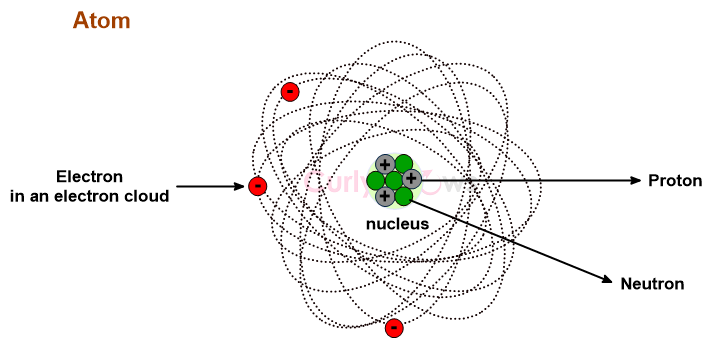
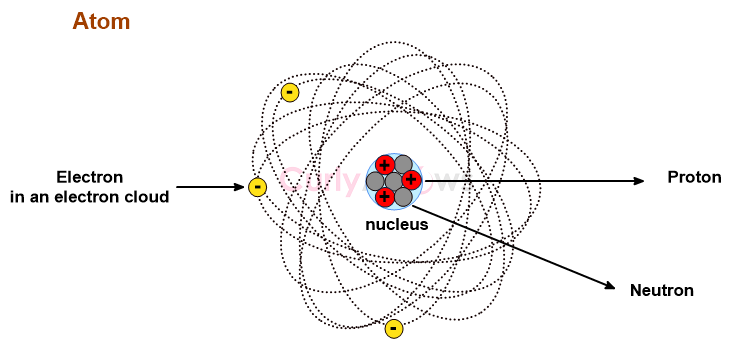
Everything around us—every breath we take, every object we touch, every star in the night sky—is built from atoms, the invisible building blocks of matter. These microscopic entities, far too small to see with the naked eye, hold the key to understanding the universe at its most fundamental level. Atoms are more than just scientific curiosities; they are the foundation of chemistry, physics, and biology, governing the interactions that form molecules, materials, and life itself.