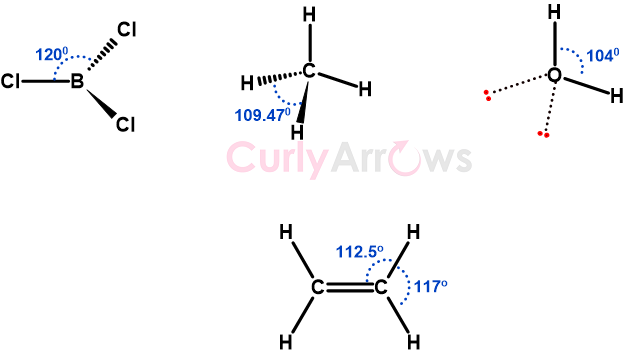
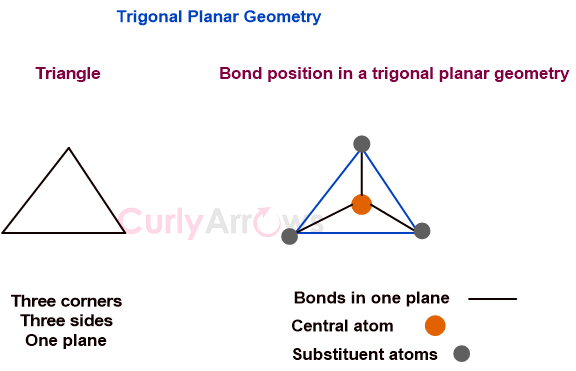
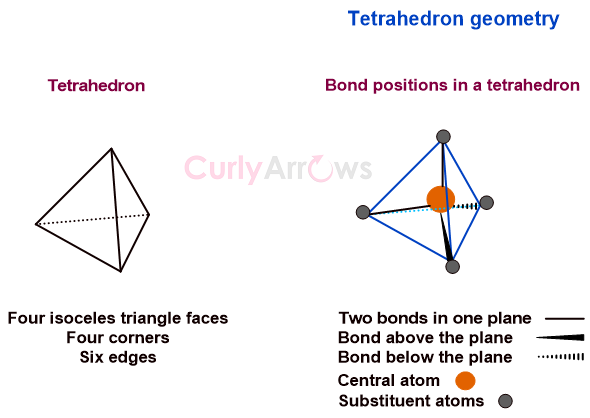
sp³ Carbon Explained: The Secret Behind Diamonds & DNA!
Imagine a single atom responsible for every organic and a few inorganic elements you see on Earth! Diamonds, methane, and even the molecules in your body. That’s sp³ Carbon—and in the next few minutes, we will see what it is and why it’s chemistry’s ultimate VIP!”
A Carbon atom can build everything from the fuel in your car to the hardest gem. The secret is in Carbon’s sp3 superpower.