A sp3 hybridized carbon is a tetravalent carbon that forms four single covalent bonds with itself or atoms of other p-block elements to its right, namely Oxygen, Carbon, Nitrogen, and Halogens. It also forms a bond with elements capable of forming covalent bonds, such as hydrogen.
The bonds formed are of equal strength and at an angle of 109.5o due to which the central carbon atom is tetrahedral in shape. Example, carbon of an alkane or an alkyl group.
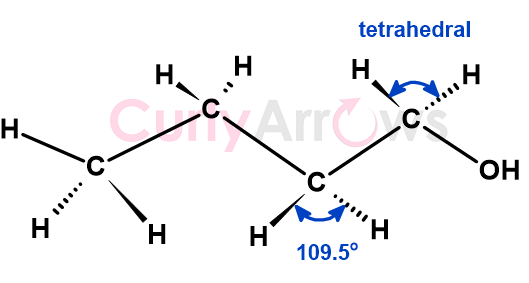
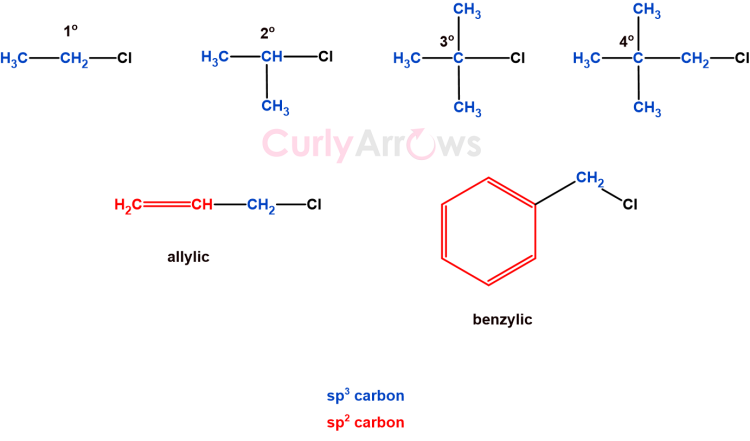
An sp3 carbon depending on the number and the type of neighbor can be further be classified as primary (1o), secondary (2o), tertiary (3o), quaternary (4o), allylic and benzylic.
Read more about sp3 hybridization of Carbon
