Conformation
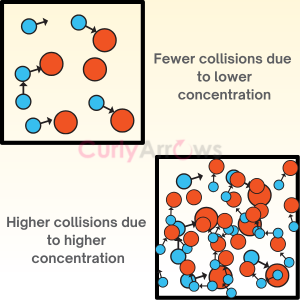
The different spatial arrangements organic molecules adopt due to the rotation of the single bond are called conformations. A specific conformation is called a conformer or conformational isomer.
Many such conformational isomers are undoubtedly interconvertible by single bond rotations. So, these isomers are simply different rotational (or structural) arrangements of the same molecule.