Electron
An electron is a negatively charged elementary particle that constitutes an atom, denoted as e- or β-. The electrons are present in the outer-nuclear region of an atom as clouds.

An electron is a negatively charged elementary particle that constitutes an atom, denoted as e- or β-. The electrons are present in the outer-nuclear region of an atom as clouds.

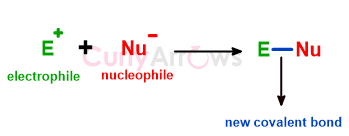
Electrophiles are electron-deficient species that accept electrons from other electron-rich counterparts, the nucleophiles, to form a two-electron covalent bond.
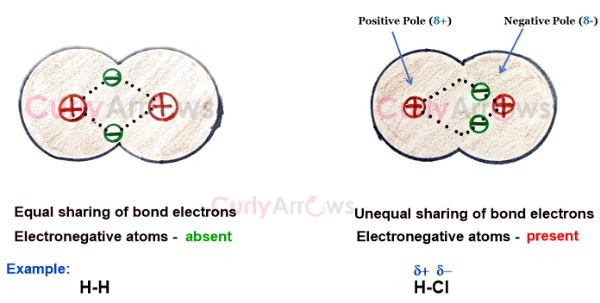
Electronegativity measures on a scale of 0.8 – 4 an atom’s or group of atoms’ tendency to attract the bond electron pair towards itself, thereby creating partial negative (δ-) and positive (δ+) terminals.
Depending on the nature of the charge (positive or negative) on the colloidal particles, its movement towards the electrode (cathode or anode) under the influence of an applied electric field is known as electrophoresis.
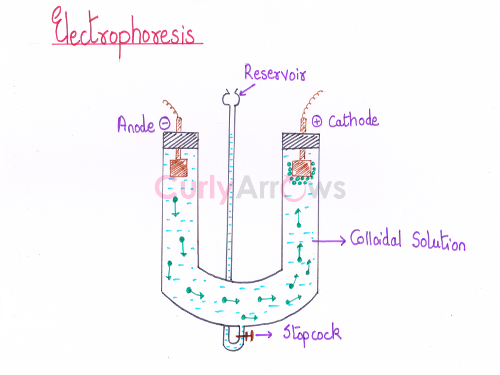
Electrophoresis proves the existence of charge on the colloidal particles and is used as a separation technique.
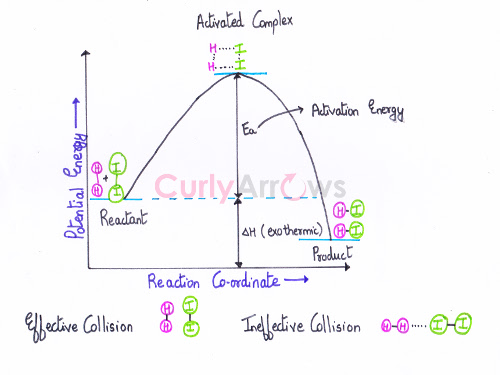
Effective collisions result in product formation due to an increase in the rate of a chemical reaction. It occurs when the two reactant molecules are correctly oriented and have attained the threshold value (or the activation energy value) at the time of the collision.
An electrode in contact with an electrolyte solution of same ionic nature (for example; Cu electrode in CuSO4 solution, Zn electrode in ZnSO4 solution) tends to either undergo oxidation or reduction due to which there develops a charge separation creating a potential difference.

In the electrochemical series, the electrodes are arranged in the increasing order of their reduction potential under standard conditions of 1M electrolyte concentration, 298 K temperature, and 1 bar atmospheric pressure.
It is also known as the activity series as the activities of two different electrodes towards displacement reaction are compared; whether the electrode under standard reaction condition will have reduction potential or oxidation potential.
A branch of chemistry that deals with the interconversion of chemical energy and electrical energy taking place via the redox reactions.
It studies how a spontaneous redox reaction capable of generating chemical energy converts it into electrical energy. Example, Electrochemical cell.
Or how the use of electrical energy can bring about a non-spontaneous chemical reaction. Example, Electrolytic cell.