Breadcrumb
- Home
- Organic Chemistry - Drawing Structures, Concepts, and Examples
- Bonding In Atoms
When atoms of different types combine to form molecules, it is a heteroatomic molecule. For example, when Carbon (C) and Oxygen (O) atoms combine under an appropriate reaction condition, it can lead to the formation of two types of products (carbon monoxide, CO, and carbon dioxide, CO2) containing two types of atoms.
A heteroatomic molecule can be diatomic (like hydrogen chloride, HCl), triatomic (like water, H2O), or polyatomic (like methane, CH4).
When the atoms combining to form molecules are of the same type, it is a homoatomic molecule. For example, when two Hydrogen atoms (2H) combine under an appropriate reaction condition, a Hydrogen molecule (H2) is formed.
A homoatomic molecule can be diatomic (like elemental hydrogen, H2), triatomic (like ozone, O3), or polyatomic (like sulphur allotrope, S8).
Atoms generally form bonding arrangements that give them filled shells of electrons like a noble gas configuration. The stability the atom aims for is that of its nearest noble gas.
For example, Lithium (Z = 3, Electronic Configuration = 1s2, 2s1) of the second row would prefer losing one electron to become Li+ (Z = 3, Electronic Configuration = 1s2) having an electronic arrangement similar to its nearest noble gas Helium (Z = 2, Electronic Configuration = 1s2).
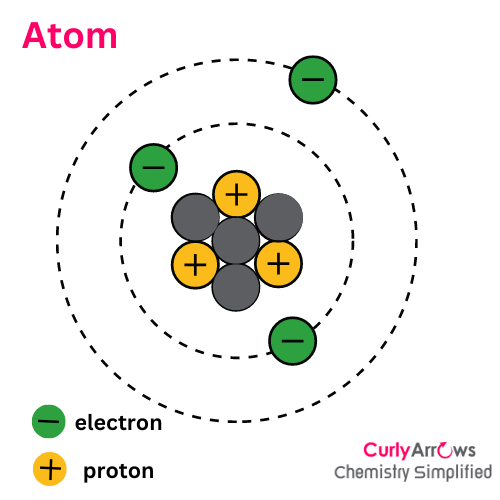
Atom, the omnipresent particle that builds the universe, hides its identity in a tiny, sub-atomic particle- the proton, where the proton number decides the type of the atom. However, an outer-nuclear component- the electrons- determines an atom's reactivity.
In nuclear reactions, the proton number can change so that the atom's identity also changes. However, in organic chemical reactions, only the electron count changes without affecting an atom's identity.
Learning Objective: To study the octet rule by G.N. Lewis, considering both ionic and covalent bonding, with examples.
Skill Level – Intermediate
Prerequisites:
Anion | Cation |
|---|---|
A negatively charged ion is an anion. Examples: Cl-, O-2 | A positively charged ion is a cation. Examples: Li+, Mg+2 |
A neutral atom's gain of electron(s) forms an anion (reduction reaction). |
Ion is different from an atom since an atom is an electrically neutral specie with an equal number of positive protons and negative electrons.
Learning Objective: To study the limitations of the octet rule, with examples.
Skill Level – Intermediate
Prerequisites:
d-orbitals
A metal atom is large, so the nucleus cannot attract and hold its outermost electrons. The metal loses those electrons and becomes positively charged kernels. The electrons then flow in the solid structure passing between these positive kernels.
.gif)
Metallic Bond | Ionic Bond |
|---|---|
| Metals have metallic bonding in them. Ex: Na, K. | Metals and nonmetals participate in forming the Ionic bond. Ex: NaCl, KBr. |
Several metal atoms lose valence electrons and become positive kernels holding a fixed lattice position. The lost electrons then freely float between the positive kernels to make the structure electrically neutral. |
In ionic compounds, a metal atom loses an electron, forms a cation, and another nonmetal atom gains it, creating an anion. Thus, there is a complete transfer of electrons between the two atoms. The ions are part of a fixed crystal lattice and immobile in the solid state. For example, NaCl, KBr.
Metallic solid consists of several metal atoms of the same kind bonded together closely. Due to their large size, metals easily lose their outermost valence electrons. The delocalized valence electron drifts and is now available for the other nuclei of metal atoms. Several delocalized electrons are comparable to a sea of electrons.
Several characteristic properties of the metals are due to the nature of the metallic bond. Some unique properties include-
1. High Melting and Boiling points:
A metallic bond is a chemical bond seen in metals consisting of tightly bound metal atoms of the same type.
Metals are large atoms that do not firmly hold their outermost valence electrons and easily lose them. Once the electrons are lost, the metal atoms become positively charged, called kernels. The position of these kernels is fixed to avoid repulsions and is part of the solid structure.
Ionic Bond | Covalent Bond | Metallic Bond | Van der Waal Forces |
|---|---|---|---|
Ionic bonds form due to the complete transfer of electrons. | Covalent bonds form due to sharing of electrons. | Metallic Bond forms between a Metal (cation) and delocalized electrons. |
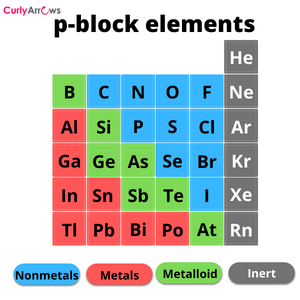
The atoms that form covalent bonds are the nonmetals (p-block elements), and they must have an electronegativity difference lower than 1.7. These values also help to classify the covalent bond into two kinds, polar and nonpolar.
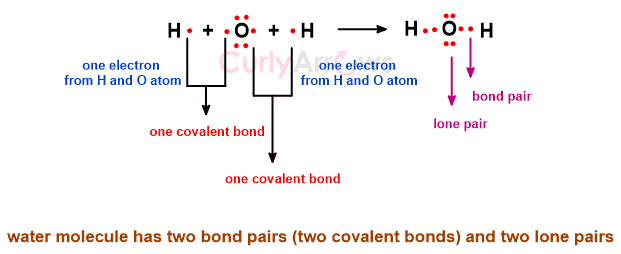
Once each electron of the two atoms forms the covalent bond, the electrons are simultaneously distributed between them so that they belong to both the atom’s nuclei. This area between the atoms is called the region of electron density.
Once the outermost valence electrons of the atoms are engaged in the covalent bond formation to make a molecule, some electrons withhold themselves from the bond-making process. This divides the electrons in the molecule into two parts- bond pair and lone pair.
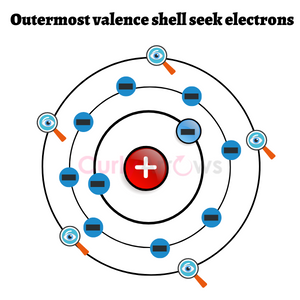
The atoms that embark on the journey of bond formation are likely to have an electron imbalance. They are less stable and become highly reactive. These atoms seek 8 electrons in their outermost shell to lower their reactivity and achieve stability.
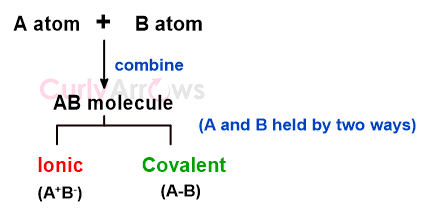
The Covalent bond is one of the two linkages by which atoms join to form molecules and is therefore classified under chemical bonds.
A metallic solid is made of many metal atoms composed of kernels and electrons, the positive kernels held in arrays while the negative electrons float around them, at the same time, attracted to each other due to their opposite electrostatic nature and engaged in bonding known as the metallic bond.
Pre-requisite: Nuclear charge, Valence electrons
Stovetop cooking routinely requires cast iron or aluminium pans but never a glass pan or a pan made of silicon. That’s because the glass pan will shatter at high temperatures, and the silicon pan will burn away.
If a two-electron covalent bond breaks symmetrically, each of the two atoms receive one electron; it is a homolytic bond cleavage.
A homolytic cleavage is shown using a fish-hook arrow, which implies one-electron movement.
A chemical bond strength is a force holding the atoms in a bond, and separating such atoms requires energy input. The bonds are of two types- Intermolecular and Intramolecular bonds.
Intramolecular bonds join the atoms in a molecule, whereas Intermolecular bonds are only responsible for closely associating the molecules. Therefore, Intramolecular bonds require higher energy to break than intermolecular bonds.