Metallic Bond | Ionic Bond |
|---|---|
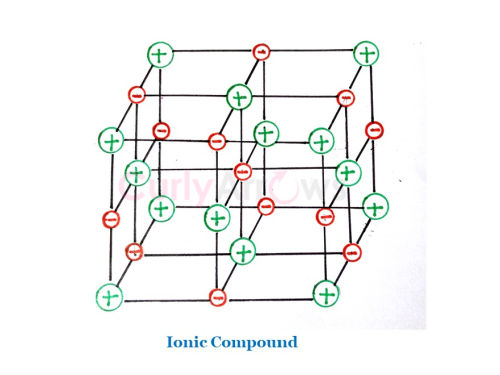
| Metals have metallic bonding in them. Ex: Na, K. | Metals and nonmetals participate in forming the Ionic bond. Ex: NaCl, KBr. |
Several metal atoms lose valence electrons and become positive kernels holding a fixed lattice position. The lost electrons then freely float between the positive kernels to make the structure electrically neutral. There is no complete transfer of electrons and the presence of cations in metallic bonding. However, metals have many free electrons. | There is a complete transfer of electrons from the metal to the nonmetal to form oppositely charged ions- a negative anion and a positive cation. |
| It is the electrostatic force of attraction between delocalized, free electrons and metal kernels. | It is the electrostatic force of attraction between oppositely charged anion and cation. |
| It is a weaker bond when compared to the Ionic bond. | The ionic bond involves ions and, therefore, is the strongest type of chemical bond, stronger than the metallic bond. |
| Metals are good conductors of electricity in a solid state. | Ionic compounds do not show electrical conductance in a pure solid state. They are conductors in the liquid state. |
| Metals are malleable and ductile because of their metallic bonds. | Ionic compounds are non-malleable and non-ductile. |
 |  |
