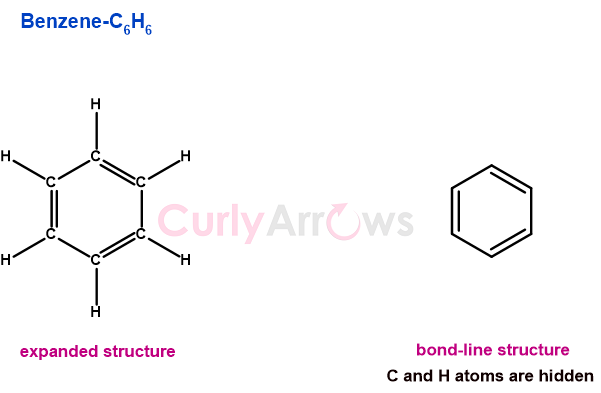
Molecular Formula
A molecular formula expresses in positive whole numbers the total count of each atom in a molecule.
For example, in the molecular formula of H2O, the total count of Hydrogen atoms is 2, and that of Oxygen atoms is 1. Another example of a complex molecule, such as glucose, expressed in a molecular formula as C6H12O6, has a total of 6 carbon and oxygen atoms, and 12 hydrogen atoms.
A molecular formula is always a multiple of the empirical formula, and their relationship is expressed as,