Newman projections offer a unique outlook on an organic molecule's appearance, particularly alkanes and substituted alkanes.
In this style of observation, an onlooker imagines viewing an organic molecule three-dimensionally to study the arrangement of substituents around the carbon atoms.
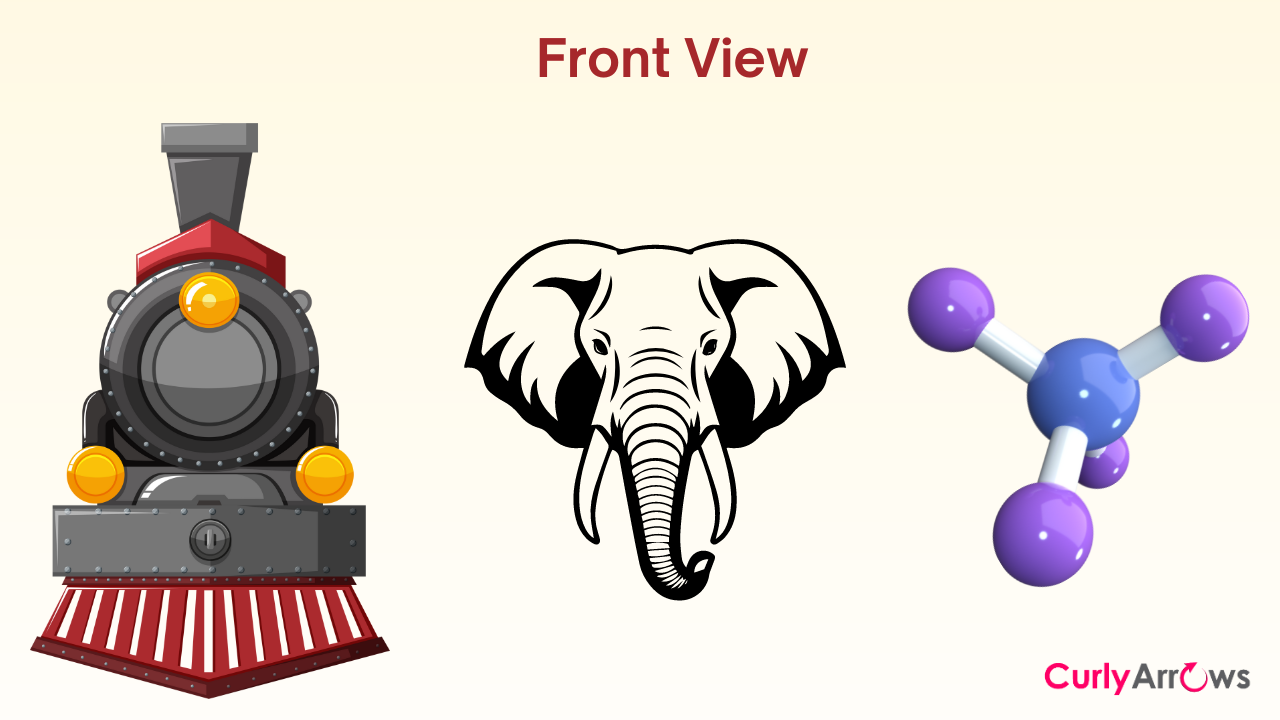
The viewer fixates his vision mainly along a Carbon-Carbon bond for drawing Newman projections. He must look straight down on the bond connecting two carbon atoms, ignoring the central connecting spine- like looking at a train from the engine side or an elephant from the trunk side while ignoring the coaches or the elephant's body. This perspective allows us to draw molecular conformations with a front-on view. It will enable the viewer to focus mainly on how the groups along the bond are distributed and their bulk.
Author's note
Besides Newman projection, the other ways to draw 3D organic molecules on 2D paper are line-wedge-dash and sawhorse projection- a different way to look at the same things.
In one-carbon molecules, the conformations don't change at all since the rotations of the single bond won't change shape; however, in molecules containing two or more carbon atoms, multiple arrangements are possible.
For example, infinite conformations are possible in two-carbon ethane due to the ease of rotation of the spinal or the central single bond. So, the angle between the hydrogen atoms in the front and back carbons can take on infinite values.
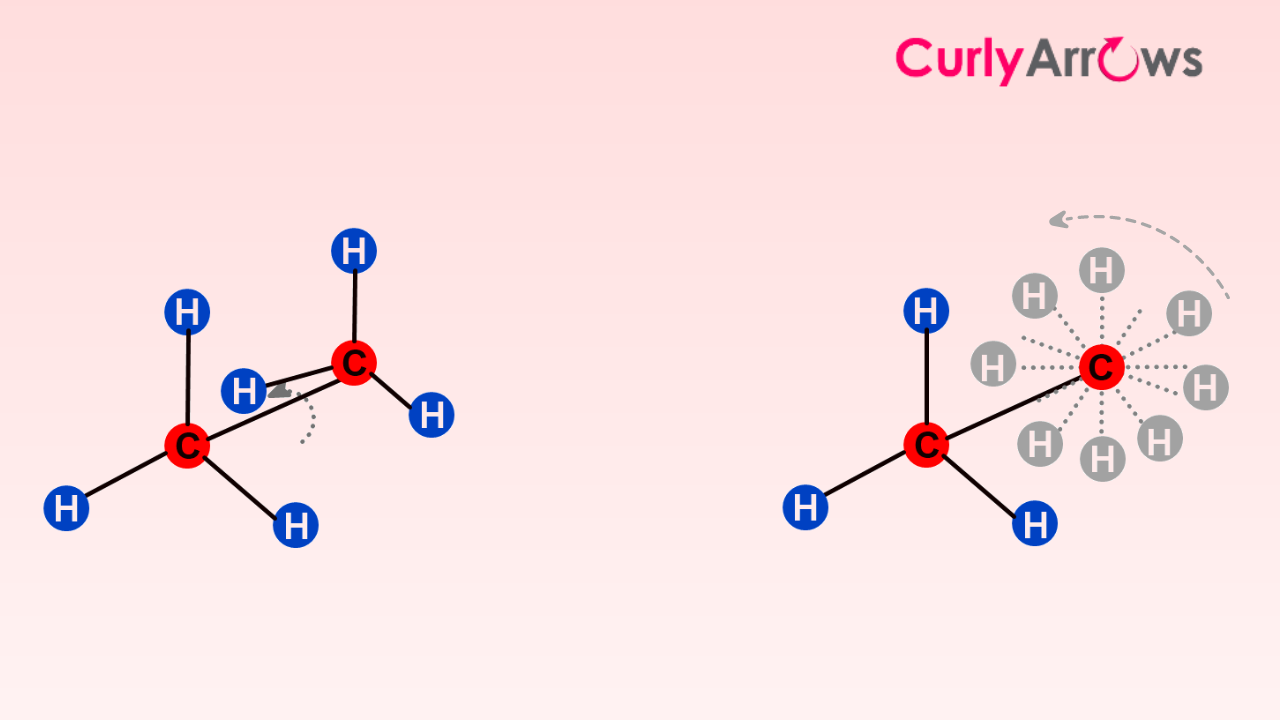
These changing conformations are represented in Newman projections using a circle and lines. The front carbon facing the viewer is represented using a dot and three lines (for three bonds carrying substituents) coming together in a Y-shape. The back carbon is represented by a circle with three bonds pointing out from the circle. (Note how each carbon's valencies are satisfied!)
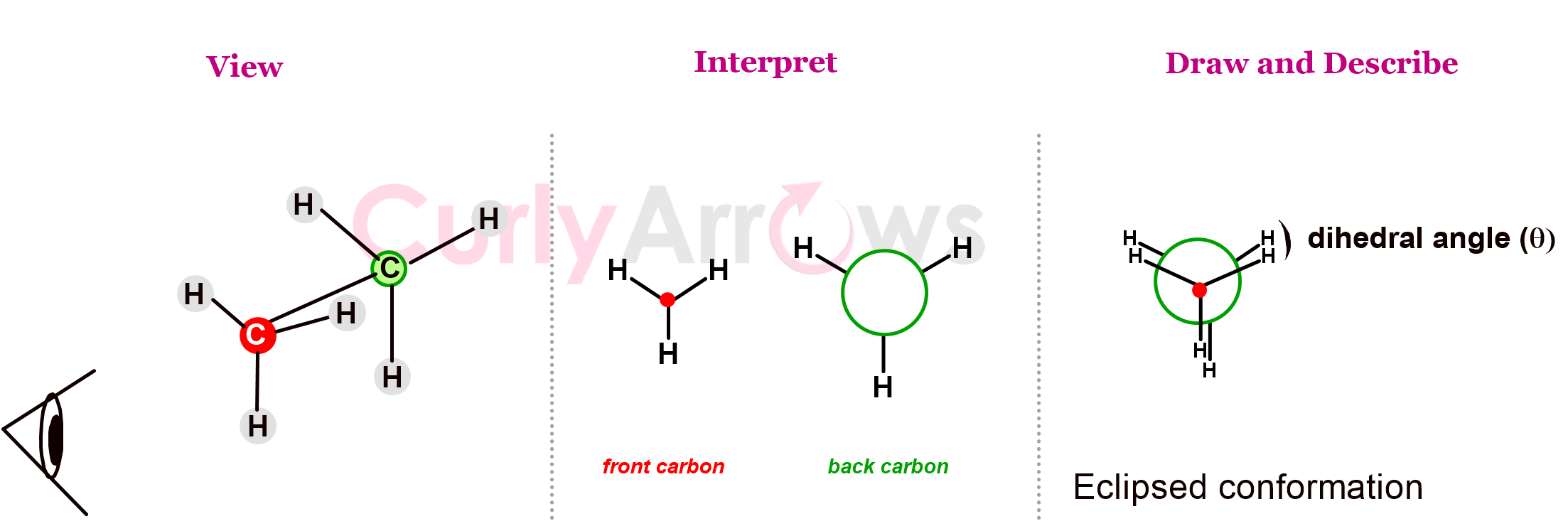
As an experiment, try holding a pen in front of your eyes and staring at it head-on. You will notice how the ends of the pen look stretched and project outwards when both your eyes are open. In Newman, we show these stretched back ends using a circle.
Any two conformations drawn using Newman projections are differentiated using the dihedral (ϑ) angle. In ethane, this is the angle between the C-H bonds on the front carbon and the C-H bonds on the back carbon.
Read Dihedral Angle
The conformations have special names. The conformation wherein the front C-H bond eclipses or hides the back C-H bond so that the dihedral angle between them is 0o, is called an eclipsed conformation. It is obtained when the C-C bond of ethane rotates by 120o, 240o, and 360o.
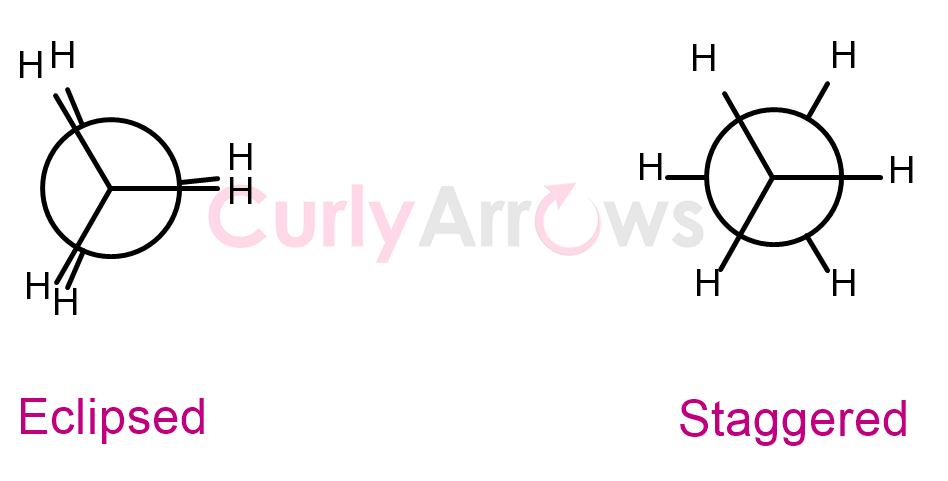
Another conformation where the hydrogen atom on the back is staggered halfway between the hydrogen atoms on the front carbon so that the dihedral angle is 60o is called a staggered conformation. The staggered conformation is obtained when the C-C bond of ethane rotates by 60o, 180o, and 300o.
Therefore, one ethane's C-C bond rotation can obtain up to six unique conformations (three staggered and three eclipsed).
All other conformations intermediate between the dihedral angle of 0o and 60o or between staggered and eclipsed conformations are called skew conformation.
While infinite conformations are possible for a molecule, it prefers those with the least interatomic repulsive interactions. This preference is driven by the molecule's potential energy, which is at its lowest when all the groups are staggered, minimizing their interelectronic repulsions.
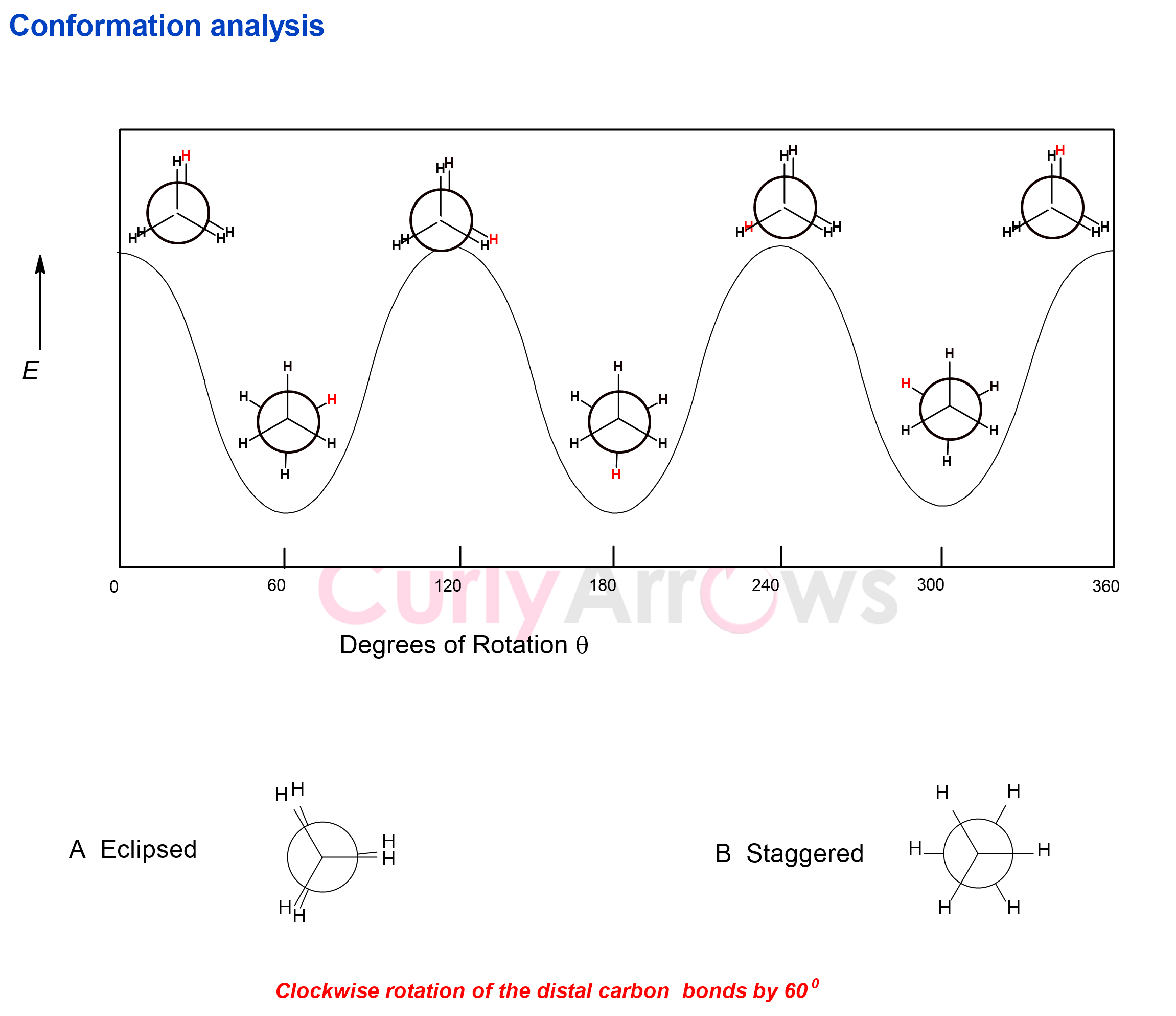
Such preferences are more likely in molecules where the second carbon atom carries a bulky group that can raise interelectronic repulsions. As the molecules go from preferred staggered to undesirable eclipse, there is an increased repulsion between the eclipsing substituents. This causes torsional strain, and the energy of the system is at its highest.
Conformations analysis is a study of the energetics of such different conformations.
In Conformations in Organic Chemistry, Chapter Stereochemistry, we have covered various stabilizing and destabilizing interactions and the nomenclature of various conformations.
Author’s Note
The Y can be drawn as rotating in several angles so you might see variations of Y like inverted and sideways. It happens when the Newman Projections are drawn where the front carbon rotates, keeping the back carbon fixed. Similarly, by keeping the front carbon fixed, the rotation of the back carbon can create projections where the circle with substituents rotates.
Since different arrangement styles may lead to different conformations that might impact a molecule's stability, drawing Newman projections enables one to propose various stable and unstable interactions that might occur as a result of that arrangement. So, by comparing and understanding the relative positions of various groups, one can conclude about the molecule's reactivity or even predict a reaction's outcome.
Next: How to Draw Newman Projections from Bond-Line Formula, 5 step-by-step solved examples on alkane, substituted alkane cycloalkane, alkene, and ketone (for subscribers)
Drawing Newman to Bond line (solved examples) (for subscribers)