Breadcrumb
- Home
- A-Z Reference Book of Chemistry Terms
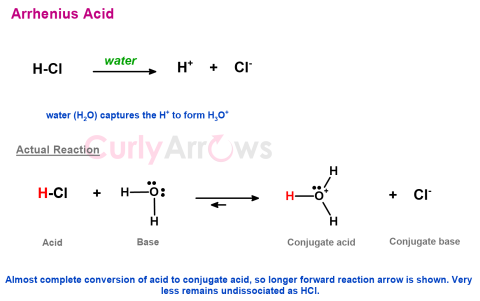 | Acid | Commonly, it is accepted that a compound is an Arrhenius acid if it liberates hydrogen ions as H+ in water. In the next step, these |
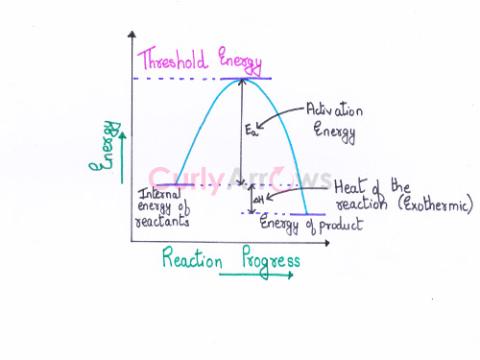 | Activation Energy (Ea) | The activation energy (Ea) is the minimum amount of extra energy absorbed by the reactant molecules from an energy source such as heat, light, etc., |
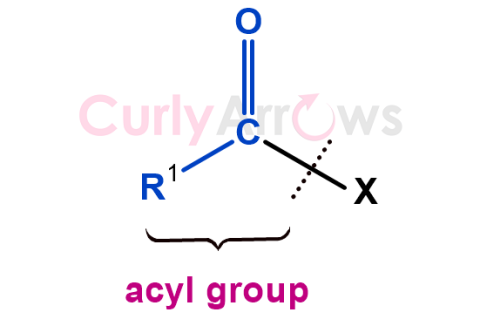 | Acyl | An acyl group is a general way of referring to an R’-C=O unit that forms the core of compounds like aldehydes, ketones, carboxy |
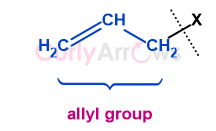 | Allyl | An allyl group is three-carbon substituted propene, also called a propenyl group (prop-2-en-1-yl).
|
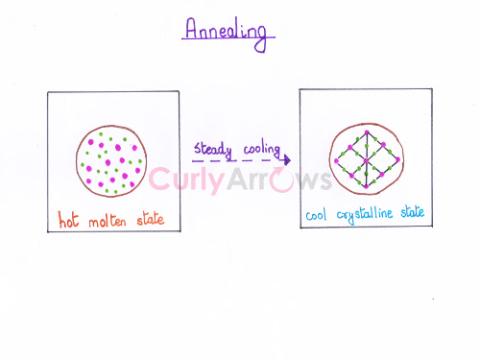 | Annealing | The process of heating a solid metal or glass to a specific high temperature and gradual cooling is called annealing. |
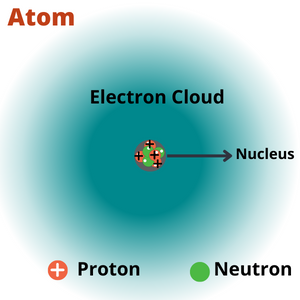 | Atom | The matter is composed of invisible, indivisible, innumerable particles called atoms. |
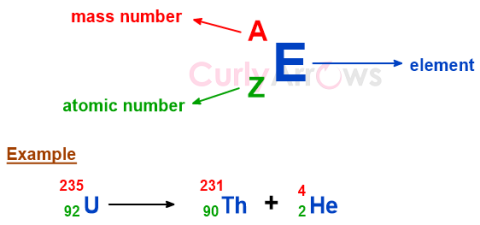 | Atomic Number | The Atomic number (symbol Z) uniquely identifies an element and is equal to the number of protons present in the nucleus of an atom. |
 | Average Atomic Mass (Atomic weight) | The Average Atomic mass of an element is the weighted average of the atomic masses of all naturally occurring isotopes of an element. |
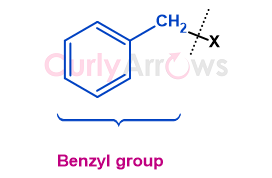 | Benzyl | A benzyl, abbreviated as Bn, refers to the phenylmethyl group (C6H5-CH2-), consisting of a benzene ring attached to a me |
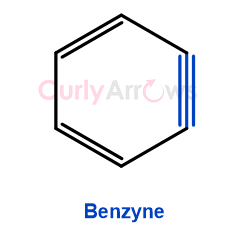 | Benzyne | Benzynes, also known as Arynes, are highly reactive intermediates formed in organic chemistry aromatic reactions. It is identified as containing a triple bond between two adjacent carbons of the benzene ring. One crucial difference separates Benzyne from Benzene... |
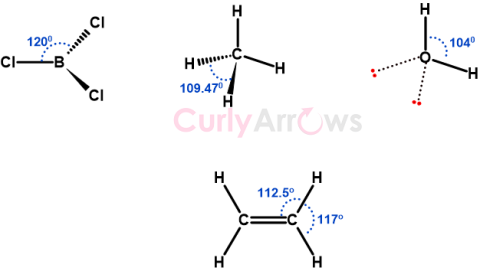 | Bond Angle | A bond angle is a geometrical angle between two bonds originating from the same central atom in a covalently bonded molecule, measured in degrees (o). |
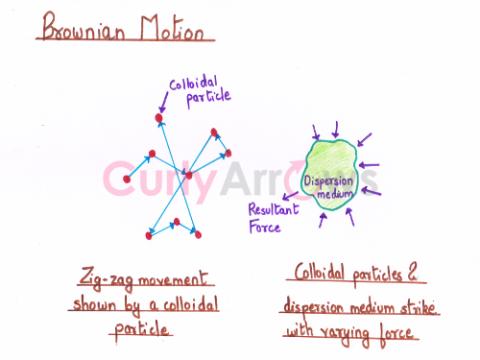 | Brownian Motion | The random collisions of the colloidal particles (dispersed phase) with molecules of the dispersion medium (colloidal sol) that results in a zig-zag movement of the colloidal particles, when observ |
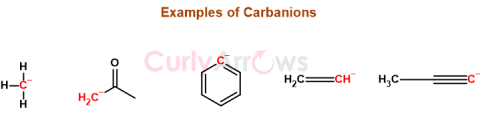 | Carbanion | A carbanion is a negatively charged, trivalent carbon ion that acts as a reactive intermediate in many organic reactions. |
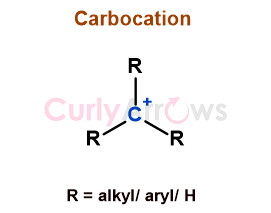 | Carbocation | A carbocation is a positively charged, trivalent carbon ion that acts as a reactive intermediate in many organic reactions. |
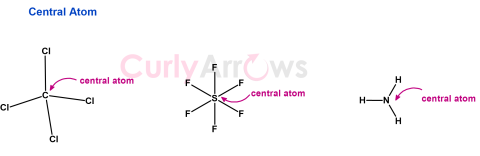 | Central Atom | In molecules consisting of more than two atoms, the least electronegative atom (except Hydrogen) is the central atom. |
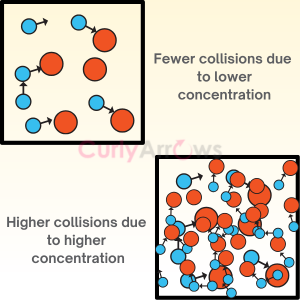 | Collision Frequency | Collision Frequency in chemical kinetics is defined as the number of collisions that take place per second per unit volume of the reaction mixture between two reactant molecules considered as |
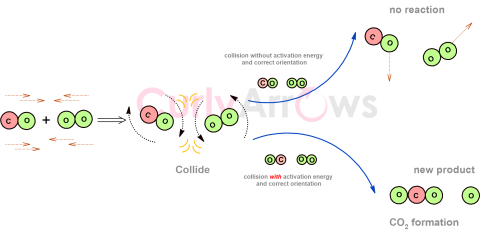 | Collision Theory | Collision theory is applicable only to bimolecular reactions mostly gases where the reactant molecules are considered as hard spheres that must collide with sufficient threshold energy and mu |
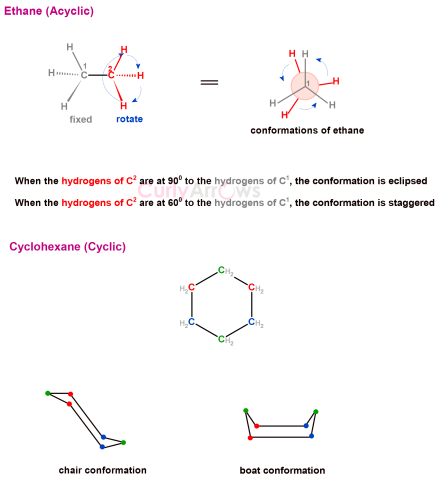 | Conformation | The different spatial arrangements organic molecules adopt due to the rotation of the single bond are called conformations. |
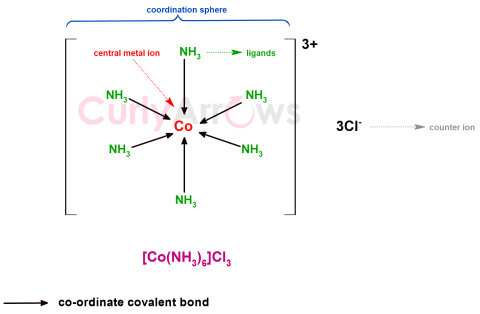 | Coordination Compounds | The compounds in which the central metal atom is linked to ligands (anions or neutral molecules) that donate its pair of electrons to form coordinate covalent bonds with the metal atom. |
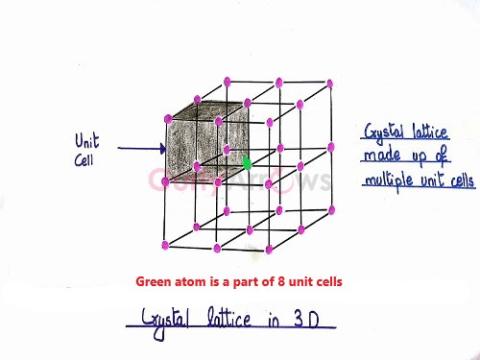 | Crystal lattice | The regular, periodic arrangement of atoms, ions or molecules at the lattice points of a crystal in a three-dimensional space is called the space lattice or the crystal lattice. |
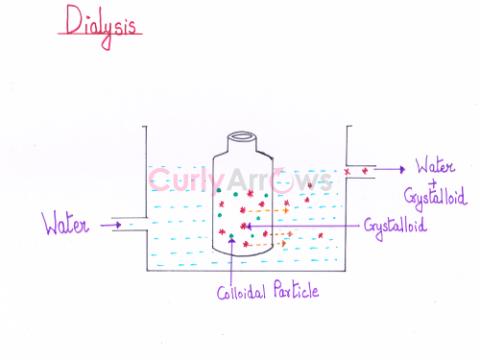 | Dialysis | Dialysis is a process of diffusion of the colloidal particles through a parchment or an animal membrane to remove excess of an electrolyte or any soluble impurities (crystalloids). |
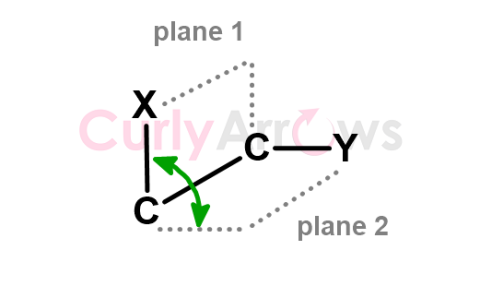 | Dihedral Angle | A dihedral angle is obtained when two planes pass through three atoms and two bonds, of which one bond is common to both planes. |
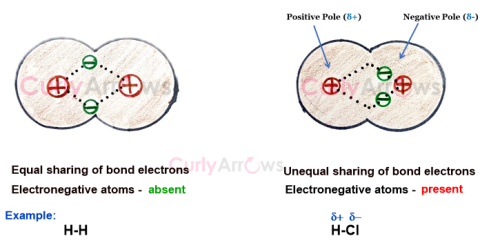
 | Dipole | The redistribution of electrons in an atom, bond, or molecule creates two ends (or poles), one electron-rich negative and the other electron-deficient po |
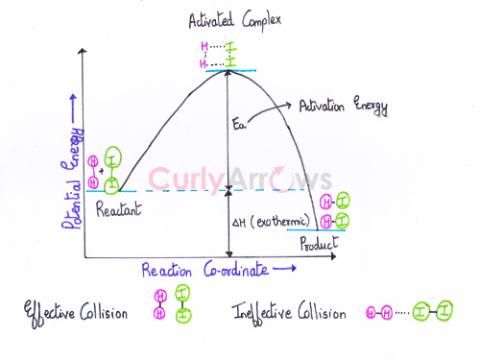 | Effective Collisions | Effective collisions result in product formation due to an increase in the rate of a chemical reaction. |
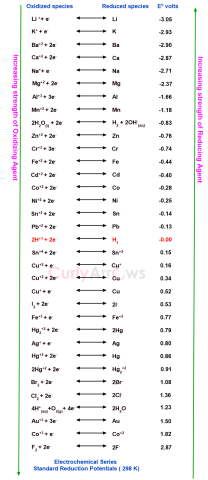 | Electrochemical Series | In the electrochemical series, the electrodes are arranged in the increasing order of their reduction potential under standard conditions of 1M electrolyte concentration, 298 K temperature, and 1 b |
 | Electrochemistry | A branch of chemistry that deals with the interconversion of chemical energy and electrical energy taking place via the redox reactions. |
 | Electrode Potential | An electrode in contact with an electrolyte solution of same ionic nature (for example; Cu electrode in CuSO4 solution, Zn electrode in ZnSO4 solution) tends to either undergo |
 | Electron | An electron is a negatively charged elementary particle that constitutes an atom, denoted as e- or β-. |
 | Electronegativity | Electronegativity measures on a scale of 0.8 – 4 an atom’s or group of atoms’ tendency to attract the bond electron pair towards itself, thereby creating |
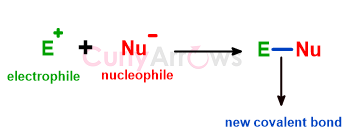 | Electrophiles | Electrophiles are electron-deficient species that accept electrons from other electron-rich counterparts, the nucleophiles, to form a two-electron covale |
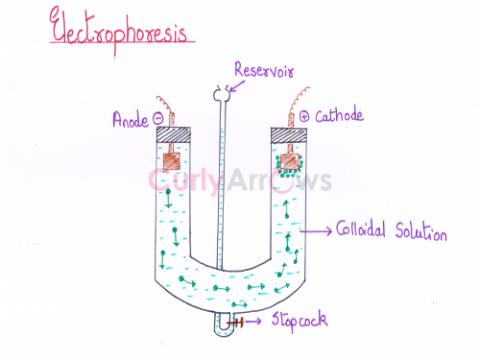 | Electrophoresis | Depending on the nature of the charge (positive or negative) on the colloidal particles, its movement towards the electrode (cathode or anode) under the influence of an applied electric field is kn |
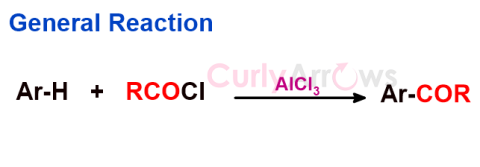 | Friedel Craft Acylation Reaction | A type of electrophilic aromatic substitution reaction to introduce an acyl group (R-C=O) in the aromatic ring resulting in aryl ketones (Ar-COR). |
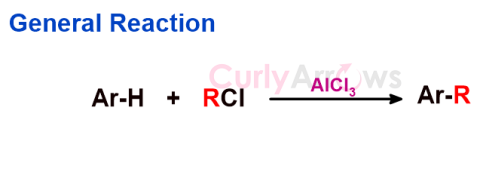 | Friedel Craft Alkylation Reaction | A type of electrophilic aromatic substitution reaction that introduces an alkyl group (-R) in the arene ring, giving alkyl arenes. |
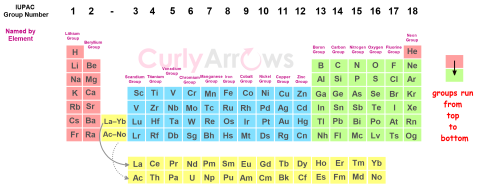 | Groups | Groups in chemistry refer to the arrangement of elements of the periodic table into vertical columns. |
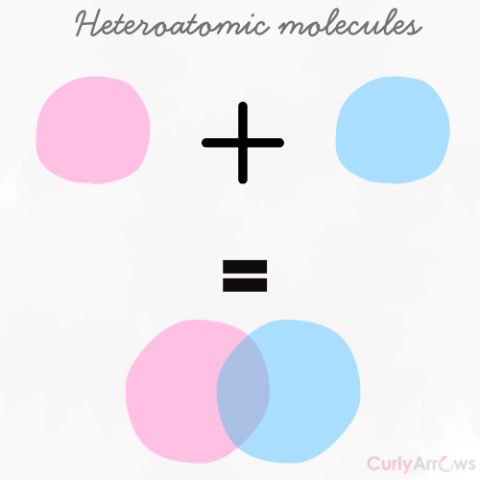 | Heteroatomic molecules | When atoms of different types combine to form molecules, it is a heteroatomic molecule. |
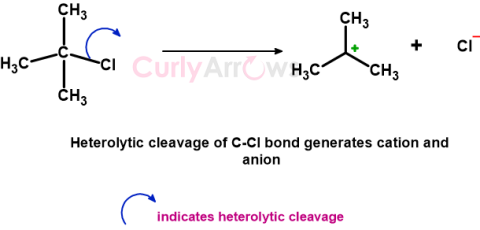 | Heterolytic Cleavage | Heterolytic cleavage or heterolysis is a chemical reaction in which the bond between two atoms breaks unequally so that the two bond electrons reside wit |
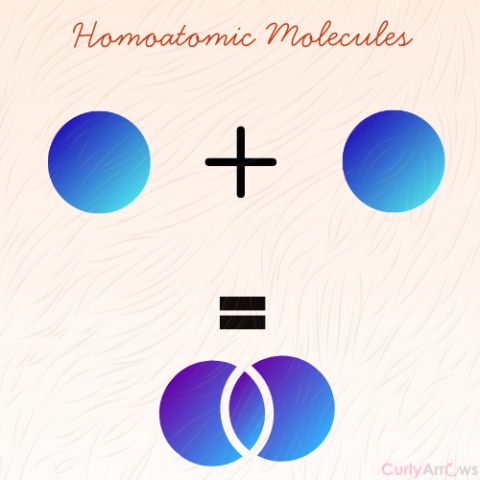 | Homoatomic molecules | When the atoms combining to form molecules are of the same type, it is a homoatomic molecule. |
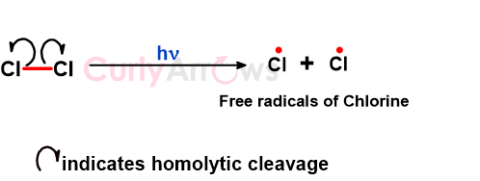 | Homolytic Cleavage | Homolytic cleavage, or homolysis, is a chemical reaction in which a covalent bond between two atoms is broken equally, and each atom retains one of the t |
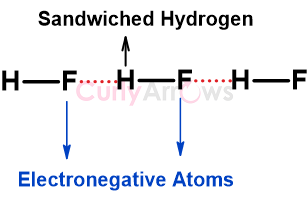 | Hydrogen Bonding | Hydrogen bonding is an attractive intermolecular interaction stronger than Vander Waals forces that exclusively occurs when the Hydrogen atom is sandwich |
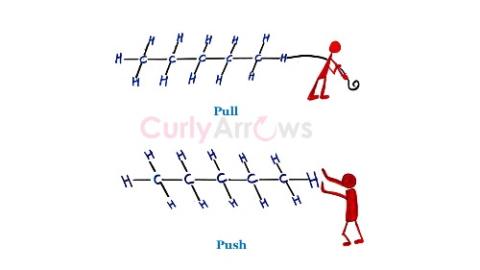 | Inductive Effect | An atom or group of atoms that can pull the bond electrons towards itself or push the bond electrons from itself and decreasingly transmit the effect alo |
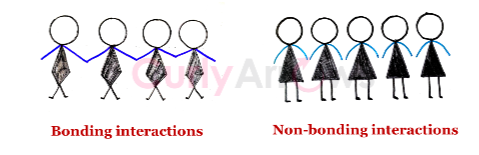 | Intermolecular Forces | Intermolecular forces are collective forces in organic chemistry that cause atoms and molecules to stick together and interact; therefore, they are electrostatic in natur |
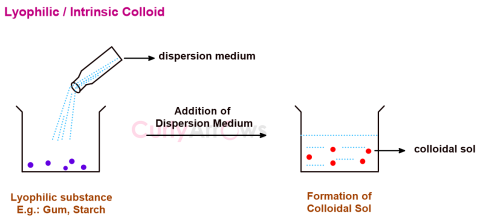 | Intrinsic Colloid | Few substances like gum, starch, gelatin, rubber have an inherent or a natural tendency to form colloidal sol on direct mixing with a suitable dispersion medium and are called intr |
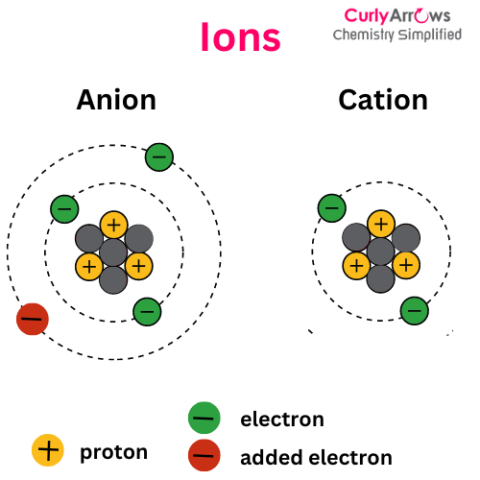 | Ion | Ion is different from an atom since an atom is an electrically neutral specie with an equal number of positive protons and negative elec |
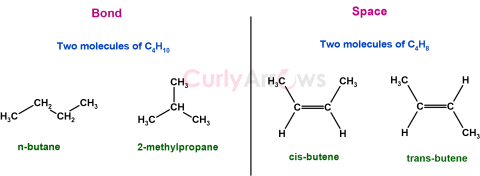 | Isomers and Isomerism | Isomers are molecules with the same molecular formula; that is, they have the same atoms in exact numbers (or the same molar masses); however, they still differ in their structures. |
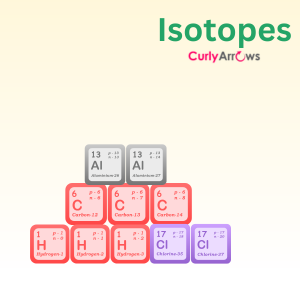 | Isotope | Isotopes are a group of atoms belonging to an element with the same atomic number but a different mass number. |
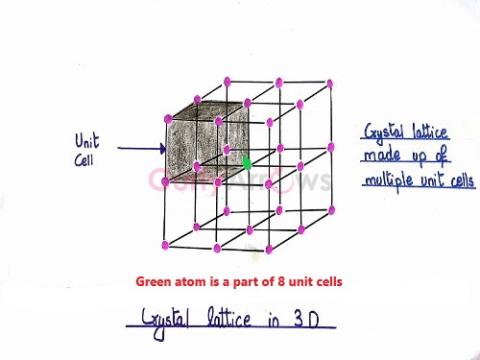 | Lattice (Chemistry) | A regular, periodic three-dimensional arrangement of atoms, ions or molecules indicating their positions in a crystalline solid. |
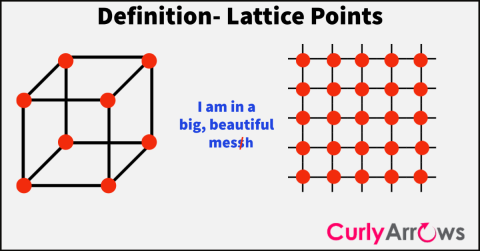 | Lattice Point | A Lattice point is the position in the unit cell or in a crystal where the probability of finding an atom or an ion is the highest. |
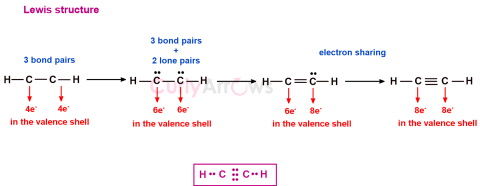 | Lewis Structures | Of all the electrons that form part of an atom, the valence electrons are the only ones that participate when atoms combine to form a bond. |
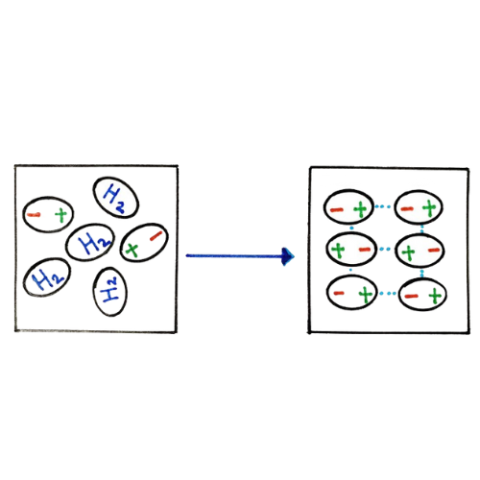 | London Dispersion Force | London Dispersion, a type of Vander Waal Force, is the weakest of the three types, yet, it is the only one universally present in all the atoms and molec |
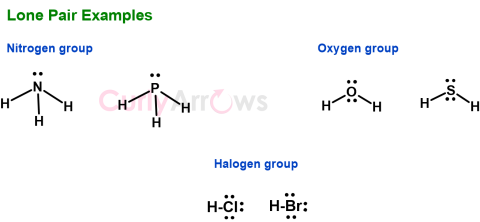 | Lone pair | Lone pair is a set of electrons present in an atom’s valence shell that did not participate in a covalent bond formation reaction; therefore, they are also called the non-b |
 | Lucas Reagent | An equimolar (1:1) mixture of a Lewis acid anhydrous ZnCl2 and concentrated HCl- Lucas Reagent, is used to identify and classify unknown alcohol (R-OH) as primary (1o), second |
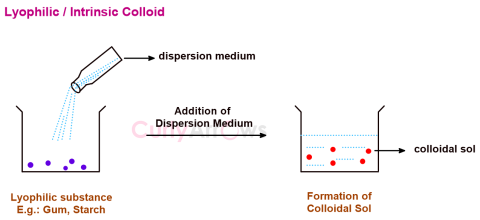 | Lyophilic Colloids | The substances that on mixing with a suitable dispersion medium readily form a colloidal sol are called lyophilic (solvent loving) and the sols th |
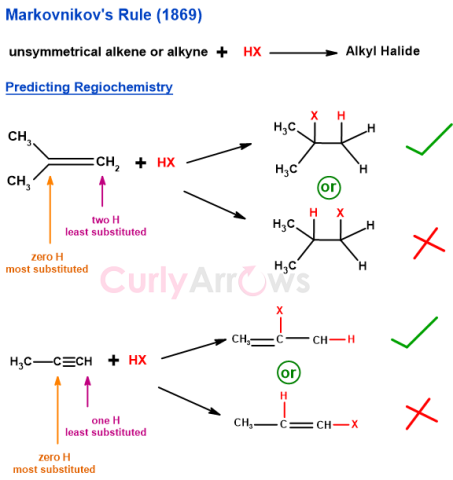 | Markovnikov’s Rule | Russian chemist Vladimir Markovnikov proposed Markovnikov’s rule in 1869 to predict the regiochemistry of addition reactions between unsymmetrical alkenes/ alkynes |
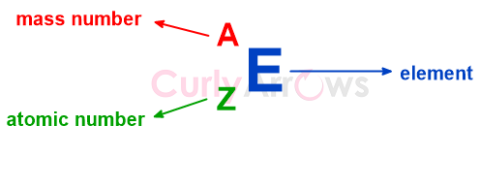 | Mass Number | The mass number is the total count of the number of protons and neutrons present in the nucleus of an atom, denoted as A. |
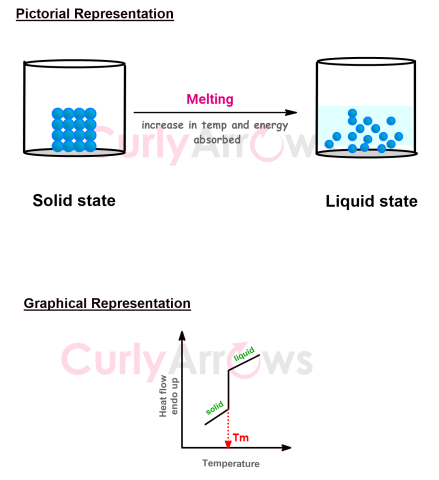 | Melting Point | The melting point of a substance is the temperature at which a solid, ordered crystalline substance is in equilibrium with a more random liquid state, thereby under |
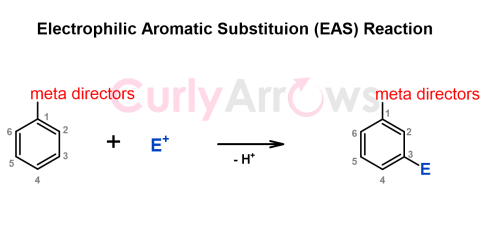 | Meta Directors | The meta directors are a class of atoms or a group of atoms that, when attached to an aromatic ring, render it with the ability to direct an incoming |
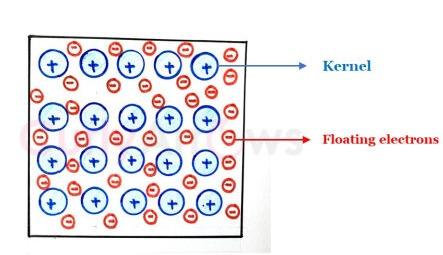 | Metallic Bond | A metallic solid is made of many metal atoms composed of kernels and electrons, the positive kernels held in arrays while the negative electrons float ar |
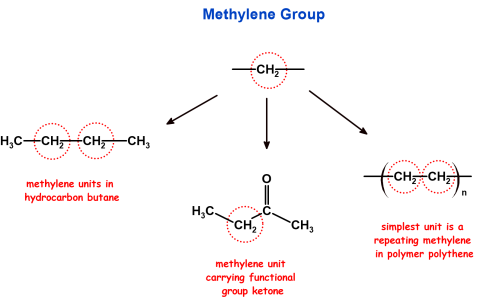 | Methylene – Species and Group | As a reactive species, a methylene refers to an electron-deficient carbene, represented as :CH2. |
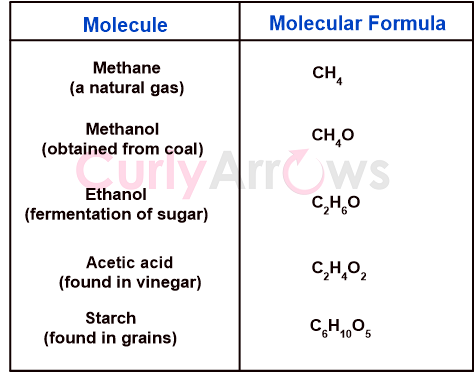 | Molecular Formula | A molecular formula expresses in positive whole numbers the total count of each atom in a molecule. |
| Molecular mass (Molecular weight) | Molecular mass is the sum of the masses of all the atoms in the molecule, denoted as m. | |
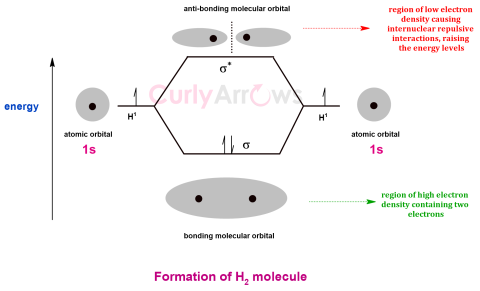 | Molecular Orbital | When atoms engage to form molecules, they do so by the overlap of atomic orbitals, creating a bigger molecular orbital that now encompasses more than one atom of a molecule. |
 | Molecule | A molecule is the smallest unit of a chemical substance that still keeps the substance's properties. It forms when two or more atoms bond together by sharing electrons. |
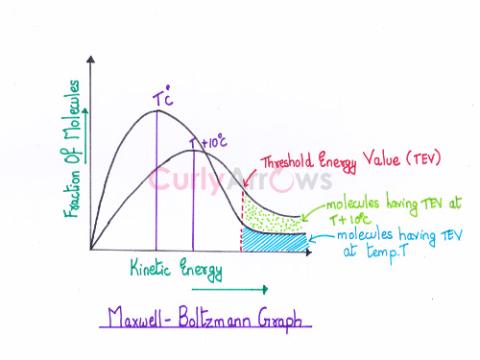 | Most Probable Kinetic Energy | The peak on the Maxwell-Boltzmann energy distribution graph (fraction of molecules versus Kinetic energy) corresponds to the most probable kinetic energy, and it is the energy possessed by the maxi |
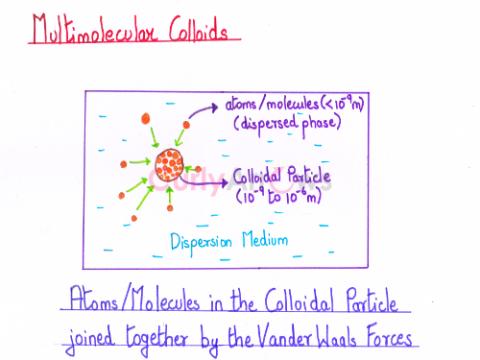 | Multimolecular Colloids | On the dissolution of a substance in a dispersion medium, the aggregation of a vast number of atoms or smaller molecules of varying sizes (< 10-9 m) leads to the formation of colloida |
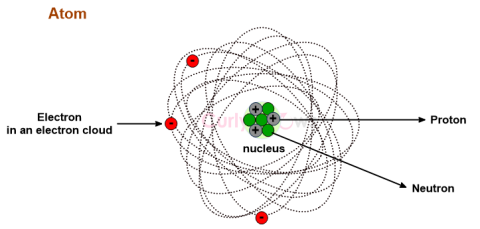 | Neutron | A Neutron is an uncharged, neutral subatomic particle found in an atom's nucleus, denoted as n or n0. |
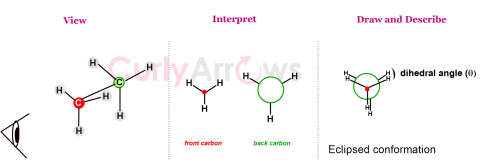 | Newman Projection | Newman Projection is a way of representing the spatial relationship of groups attached to two adjacent carbon atoms in a molecule. |
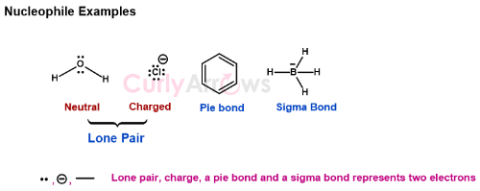 | Nucleophiles | Nucleophiles are an atom or a group of atoms that are richer by two electrons and donate these electrons to electron-deficient species, the electrophiles. |
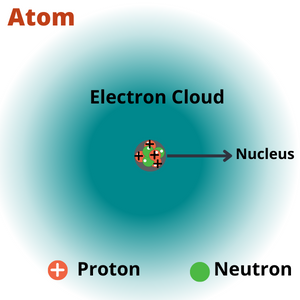 | Nucleus of an atom | An atom has a central, dense, tightly packed nucleus with a diameter of 10-15 m compared to an atom at 10-10 m, a size equivalent t |
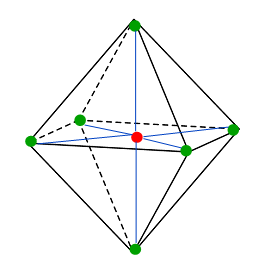 | Octahedral Geometry | When an atom is surrounded by six substituents, arranged in a manner that four are in one plane, one above and below, and their vertices join to give eight faces (oct |
 | Octet Rule | Atoms generally form bonding arrangements that give them filled shells of electrons like a noble gas configuration. |
 | Organic Chemistry | Organic chemistry is a branch of chemistry that studies transformations at three levels- macroscopic (visual changes like rotting of an apple), m |
 | Oxidation | Oxidation Reaction according to the Classical ConceptOxidation is Defined as, the addition of Oxygen or any other electronegative element to a substance. Example, |
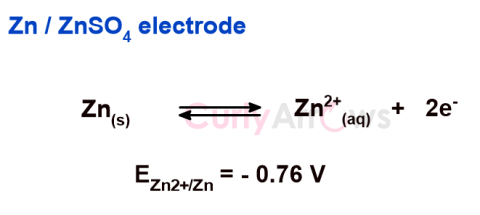 | Oxidation Potential | For an electrode in contact with an electrolyte solution having similar ionic nature, if the tendency of the electrode is to lose electrons and undergo oxidation reaction, the electrode is said to |
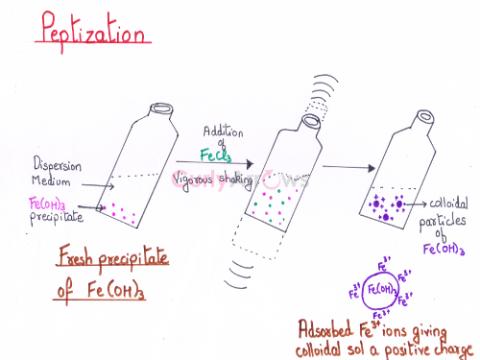 | Peptization | The process of transforming a freshly prepared precipitate to a colloidal sol by shaking it with a small amount of electrolyte having an ion common to the precipitate, in a dispers |
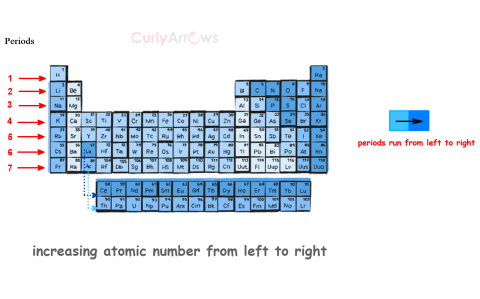 | Periods | Periods in chemistry refer to the horizontal rows of elements in the periodic table, which represent all 118 elements across 7 rows. |
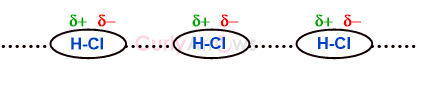 | Permanent Dipole | A permanent dipole is an inherent feature of the molecule due to the nature of the participating atoms forming the two-electron covalent bond. |
 | Physical Properties | Physical properties identify the substance's unique nature by subjecting the substance to qualitative and quantitative measurements. |
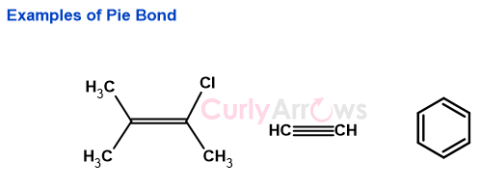 | Pi bond | Once the sigma bonds are formed, any additional electrons in the perpendicular unhybridized p-atomic orbitals engage in side-to-sid |
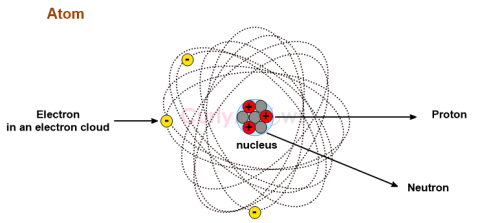 | Proton | A proton is one of the subatomic particles found in every atom’s nucleus, denoted as p or p+. |
 | Reduction (Chemistry) | Reduction Reaction according to the Classical ConceptReduction is defined as, the elimination of oxygen or any electronegative element from a substance. Example, |
 | Reduction Potential | For an electrode in contact with an electrolyte solution having similar ionic nature, if the tendency of the electrode is to gain electrons and undergo reduction, the electrode is said to have a re |
 | Resonance | Resonance theory explains various observed properties in a molecule using the electron delocalization concept and multiple Lewis structures, which a single Lewis structure cannot. |
 | Sawhorse Projection | It is a way of visualizing the carbon atoms of an open-chain organic molecule from an oblique angle and projecting that image on a 2-dimensional paper to appear like a ca |
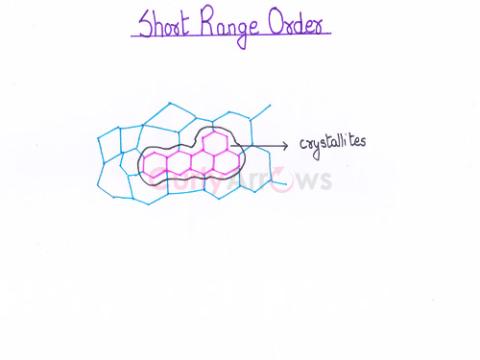 | Short Range Order | The regular periodic arrangement of the constituent particles at the lattice points of the unit cell is observed only in a short region. |
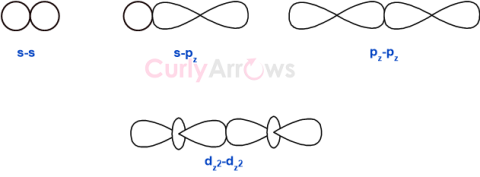 | Sigma Bond | The single covalent bond is referred to as a sigma bond, denoted by the symbol σ. |
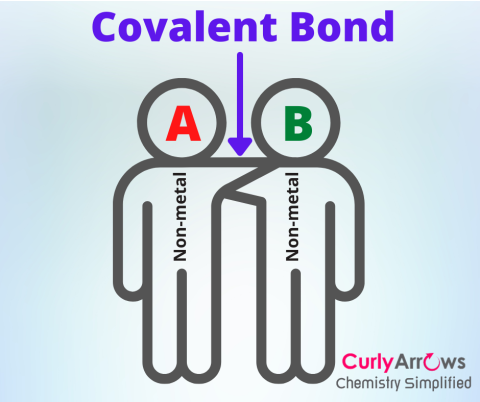 | Single Covalent Bond | When two atoms come closer by attraction and overcome their repulsive interactions until they find a balance, at which point, the atoms contribute one valence ele |
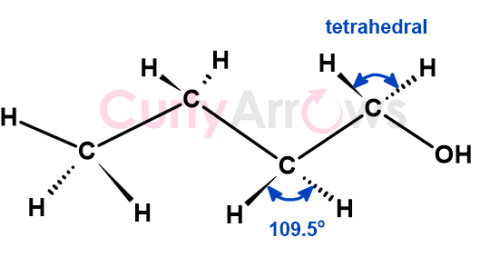 | sp3 Carbon | A sp3 hybridized carbon is a tetravalent carbon that forms four single covalent bonds with itself or atoms of other p-block elements to its ri |
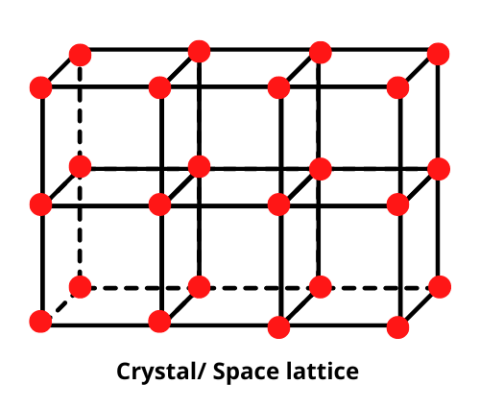 | Space lattice | The regular, periodic arrangement of atoms, ions or molecules at the lattice points of a crystal in a three-dimensional space is called the space lattice or the crystal lattice. |
 | Stereoisomers | Stereoisomers are molecules that have the same molecular formula and bonding arrangement; however, they differ in how their atoms are po |
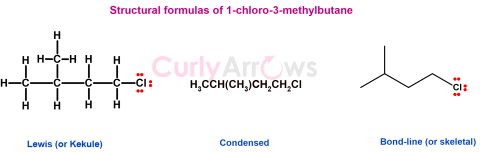 | Structural Formulas | Structural formulas are used to display covalently bonded molecules in the order they are bonded. The atoms may bond using single, double, or triple bonds. |
 | Structural Isomers | Structural isomers have the same molecular formula but differ in how the atoms are bonded to each other; that is, their attachment style is diffe |
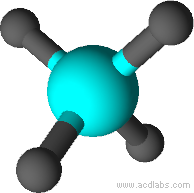 | Tetrahedral Geometry | In a regular tetrahedral molecular geometry, a central atom is surrounded by four substituents that occupy the four corners of a tetrahedron. |
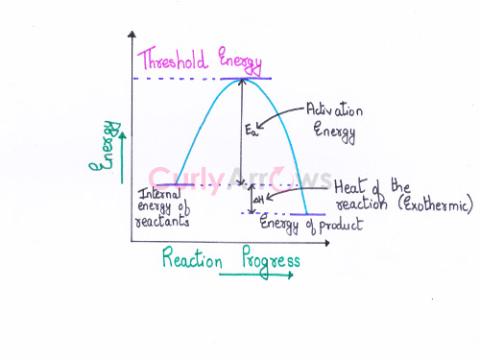 | Threshold Energy | Threshold energy is the minimum kinetic energy the molecules must have to bring about effective collisions between two reactant molecules considered as hard spheres, resulting in a chemical reactio |
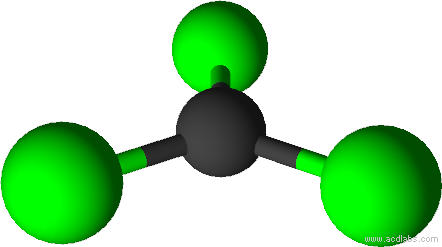 | Trigonal Planar Geometry | In a regular trigonal planar molecular geometry, a central atom is surrounded by three equally spaced substituents in one plane, so joining the three cor |
 | Unified atomic mass unit | The unit used to describe the mass of an atom is the unified atomic mass unit, symbolized as amu or μ. |
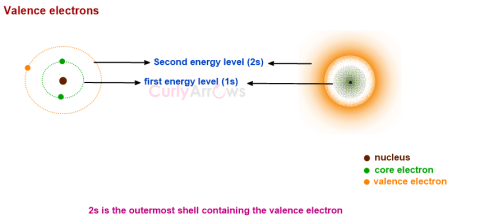 | Valence Electrons | The outermost electrons of an atom that are mostly involved in bonding reactions are called valence electrons. |
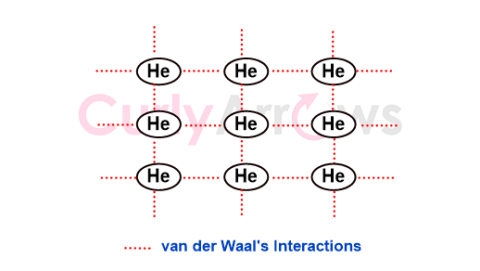 | Vander Waal Forces | Vander Waals is an attractive universal force that operates when the atoms and molecules are within a distance range of 0.4- 0.6 nm. |
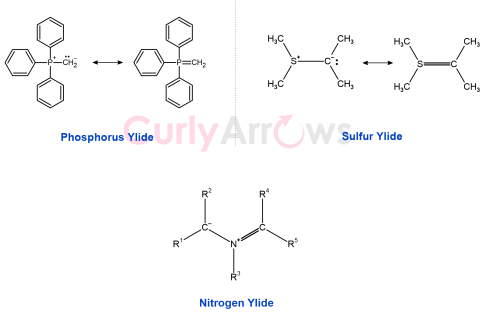 | Ylide | The ylide is an electrically neutral molecule that has a negative carbon with an unshared electron pair (usually a carbanion) adjacent to a positive heteroatom (typically |
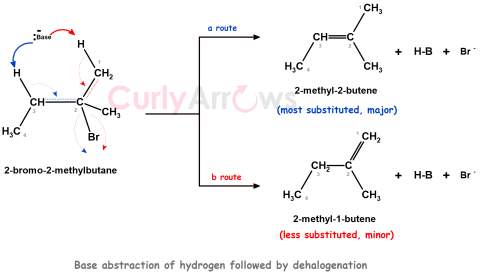 | Zaitsev’s Rule | A rule stating that an elimination reaction will give as the major product the most stable alkene, that is, the alkene with the most highly substituted double bond. |