Structural isomers have the same molecular formula but differ in how the atoms are bonded to each other; that is, their attachment style is different.
Such molecules that differ by bonds while still having the same molecular formula are also known as constitutional isomers.

Constitutional isomers are further subdivided into 6-types depending on where the variation in the structure exists.
1. If the isomers vary in the chain length, then the isomers are chain isomers. Such type of isomerism is commonly seen in alkanes, alkenes, and other organic compounds with long carbon chains.
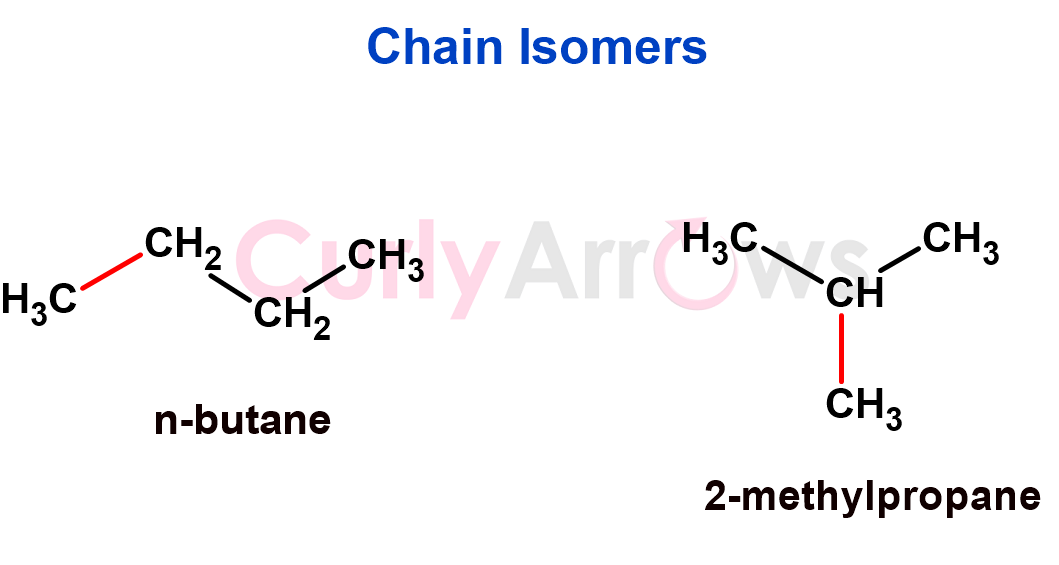
Since the change is in the hydrocarbon part of the molecule, they will have different physical properties (e.g., boiling points, melting points) due to differences in molecular shape and intermolecular forces. Chemical properties may be similar.
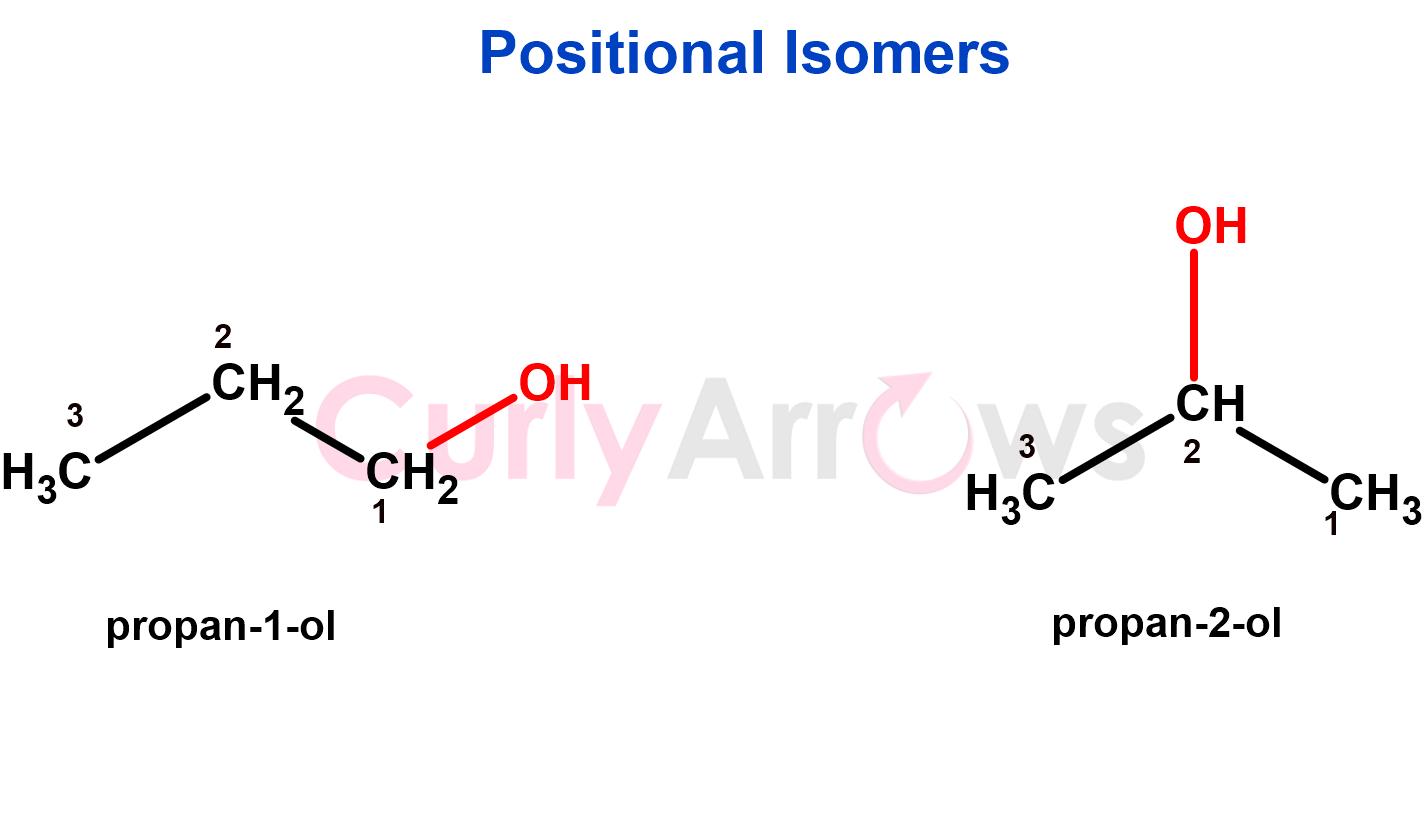
2. If the structural variation is in the position of the functional groups or multiple bonds, the isomers are positional isomers.
Since the functional group remains unchanged between the structures, these isomers will exhibit similar chemical properties but different physical properties (e.g., boiling points, melting points) due to variations in molecular shape and polarity.
3. Isomers with the same molecular formula but different functional groups are functional isomers.
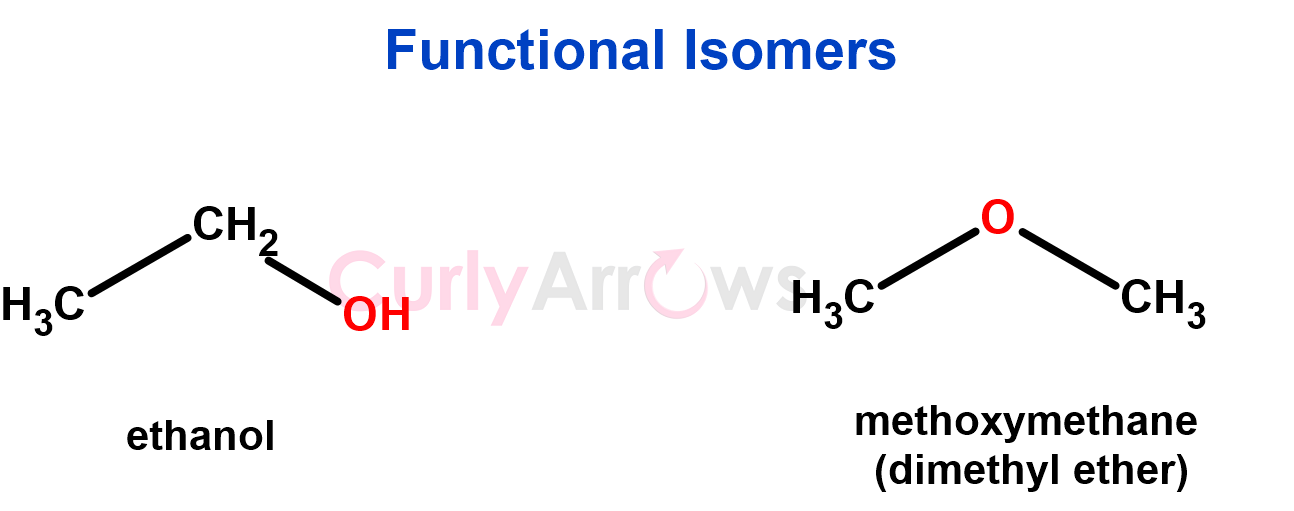
These molecules have altogether different functional groups; therefore, their chemical reactivities and behaviour would be different. They would also have slightly different physical properties due to the unique nature of their functional groups (for example, a free -OH would be a stronger H-bond participant, affecting the intermolecular interactions and subsequently the boiling point).
4. Compounds with the same molecular formula but different carbon atoms on either side of the functional groups are metamers.
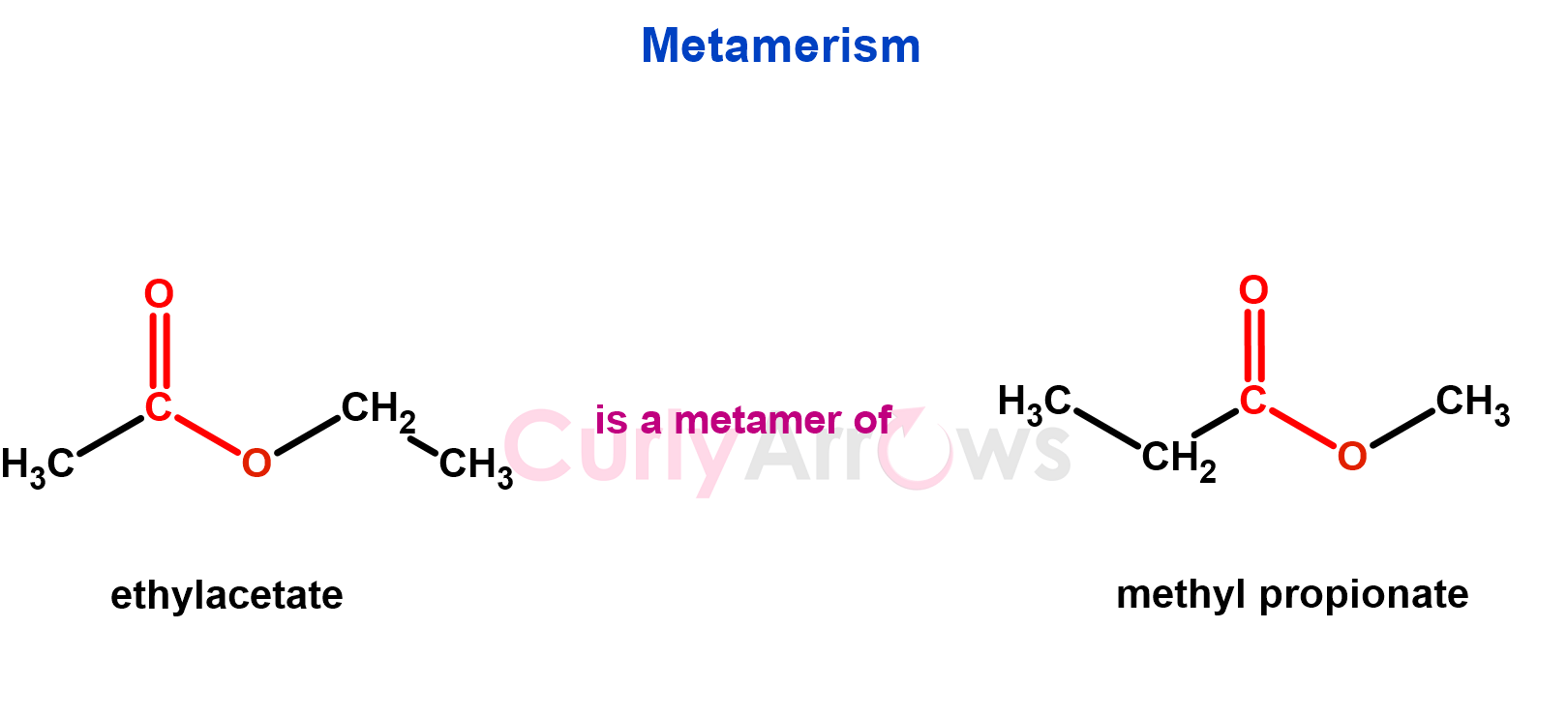
Metamers have matching functional groups; therefore, they exhibit similar chemical properties but differ in physical properties (e.g., boiling points, solubility) due to variations in their carbon chain structures.
5. Functional group isomers wherein the two isomers rapidly interconvert due to H-atom migration are called tautomers. This can cause a shift in the position of a double or single bond, often involving a functional group like carbonyl (-C=O) or imine (-C=N-).
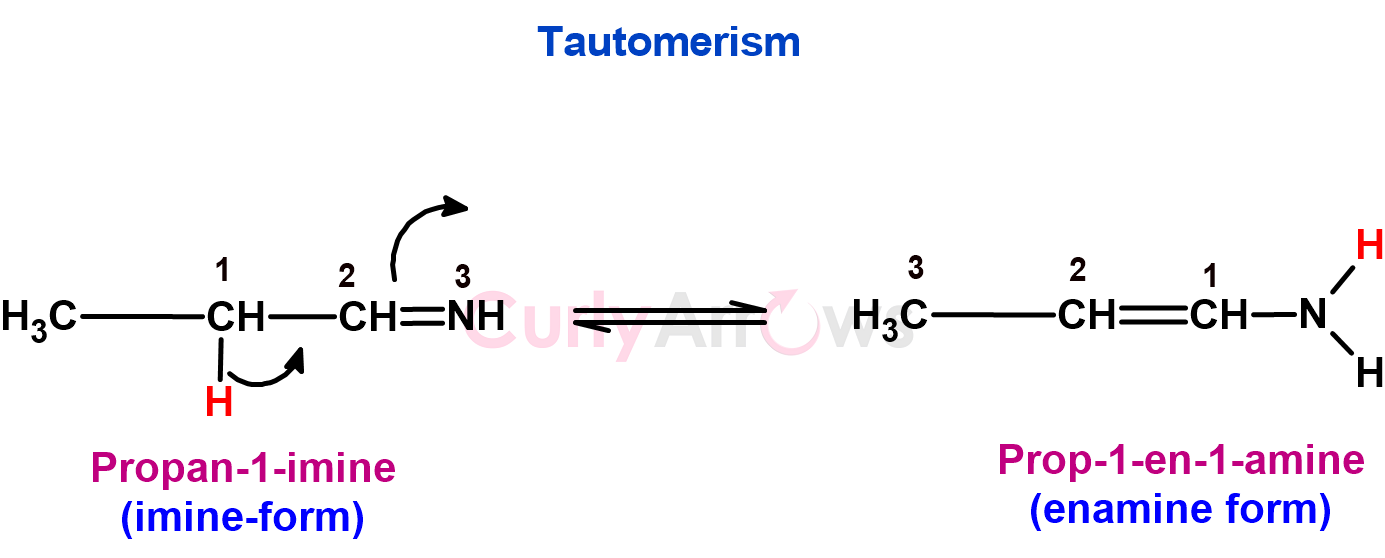
Tautomers have different structures, so their chemical and physical properties can differ, but they interconvert rapidly, making them appear as a single compound in many reactions.
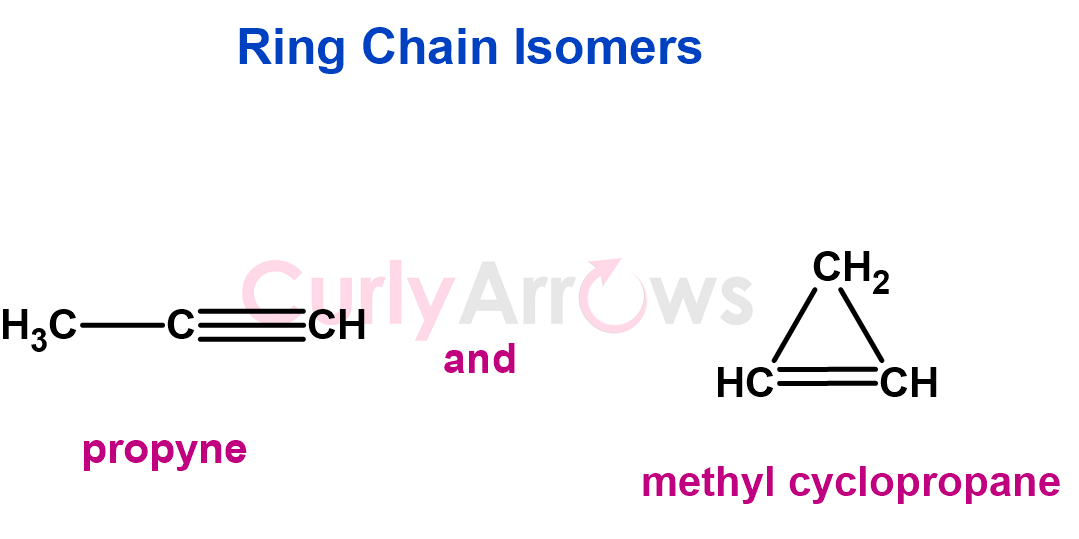
6. Compounds with the same molecular formula but that can alternate between open-chain and cyclic structures are called ring-chain isomers.