Trigonal Planar Geometry
In a regular trigonal planar molecular geometry, a central atom is surrounded by three equally spaced substituents in one plane, so joining the three corners would give a triangle.
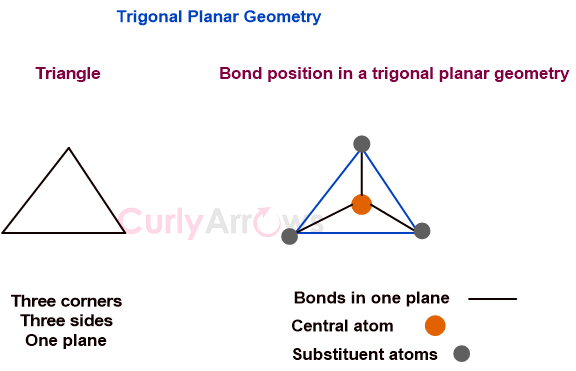
In a regular trigonal planar molecular geometry, a central atom is surrounded by three equally spaced substituents in one plane, so joining the three corners would give a triangle.

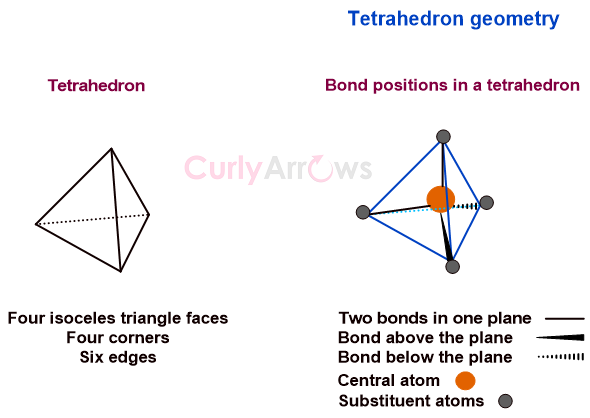
In a regular tetrahedral molecular geometry, a central atom is surrounded by four substituents that occupy the four corners of a tetrahedron. The substituents are called ligands if the central atom is a metal.
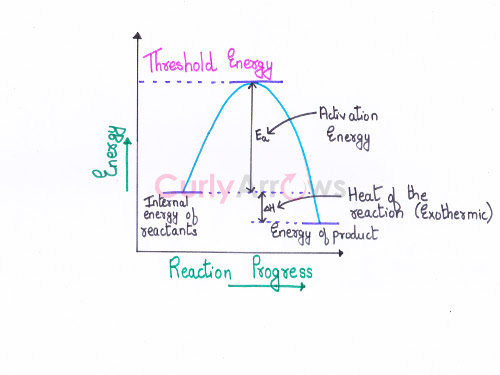
Threshold energy is the minimum kinetic energy the molecules must have to bring about effective collisions between two reactant molecules considered as hard spheres, resulting in a chemical reaction.
Therefore,
Threshold energy= Average of the initial kinetic energy possessed by the reactants + Activation energy (Ea)