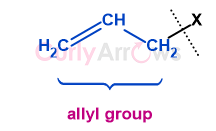
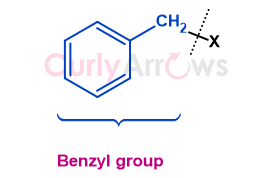
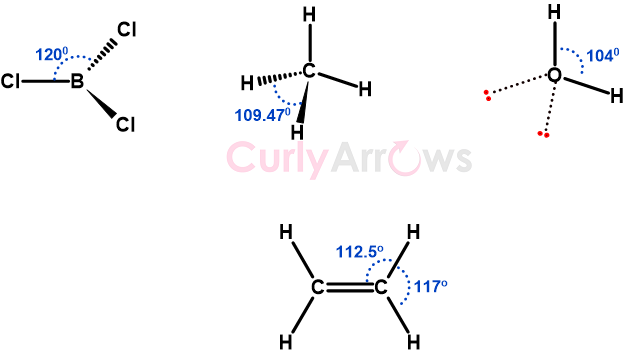
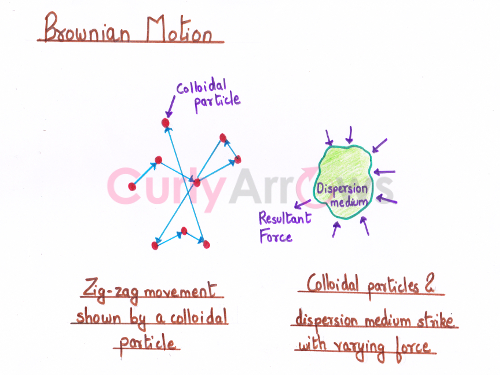
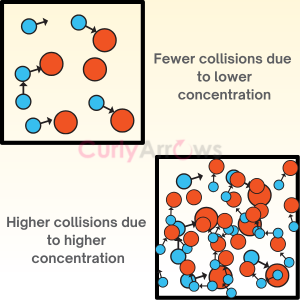
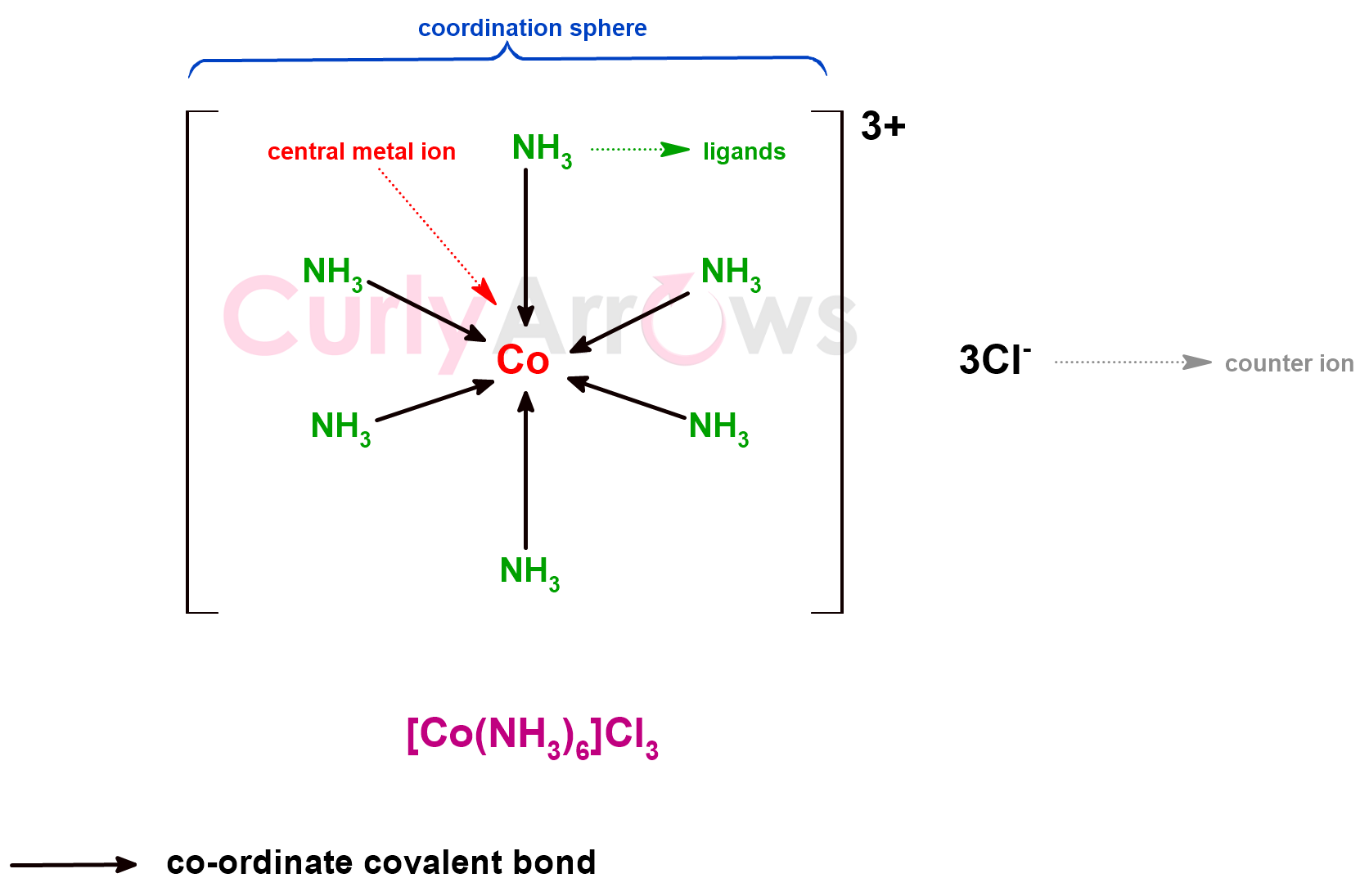
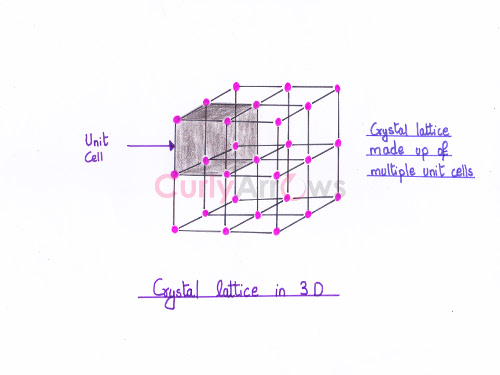
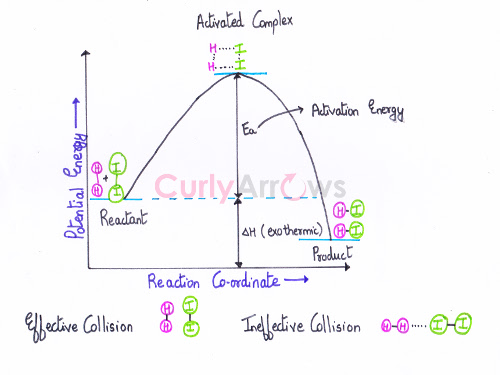
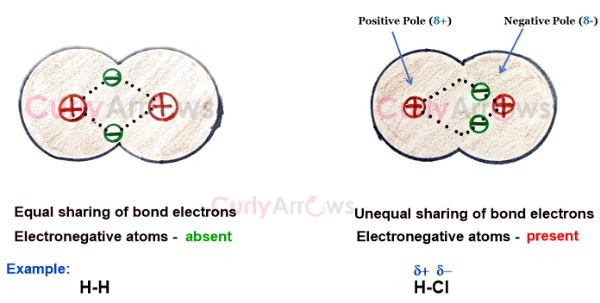
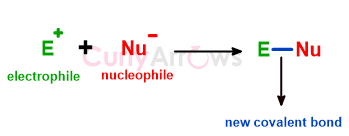
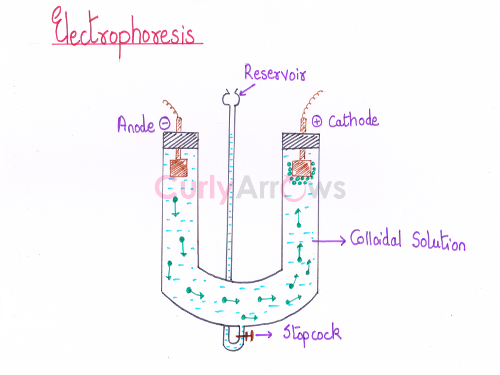
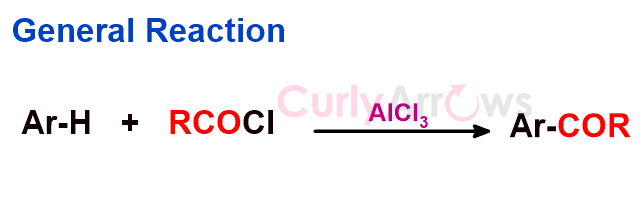
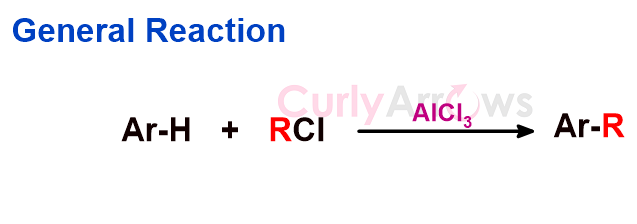
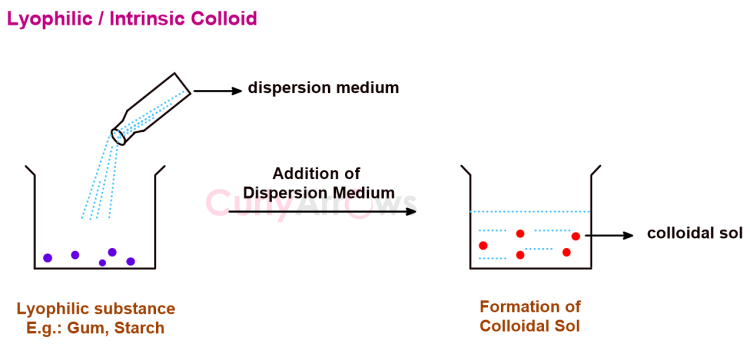
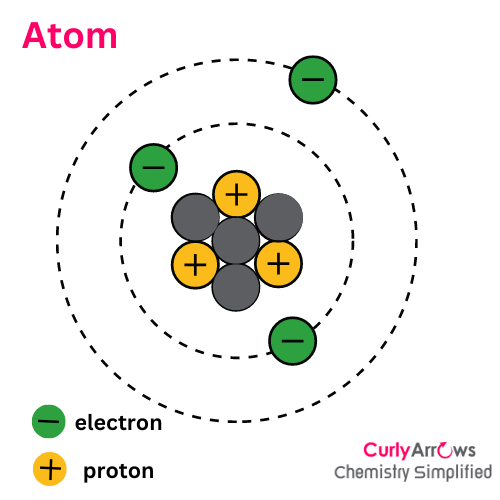
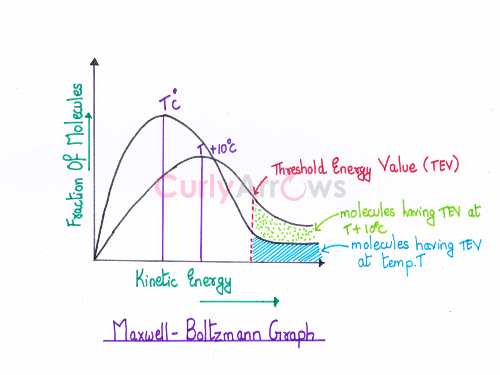
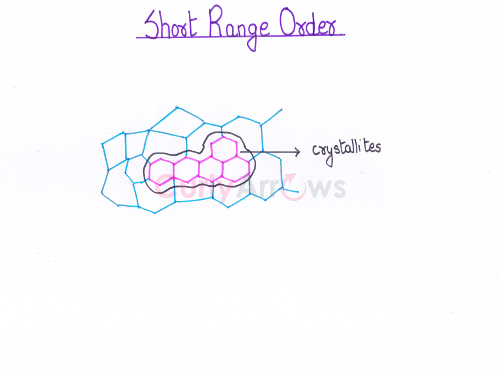
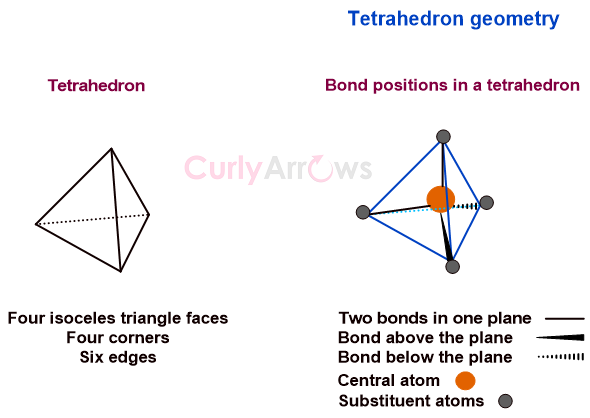
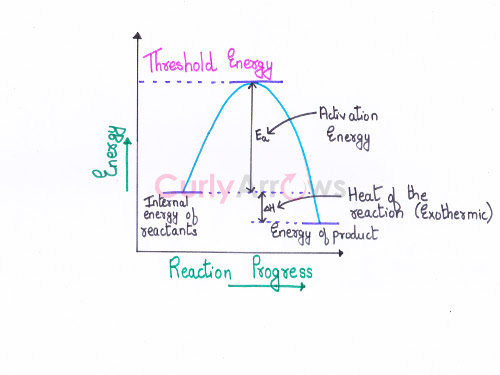
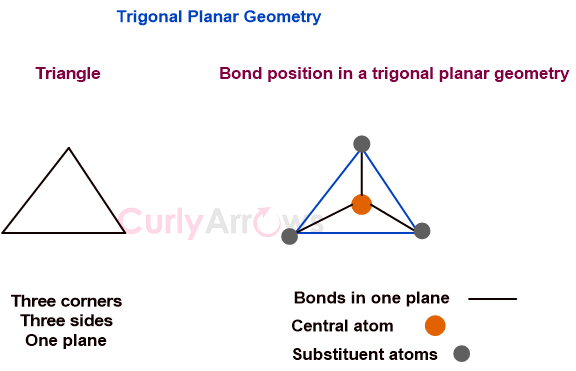
Acid
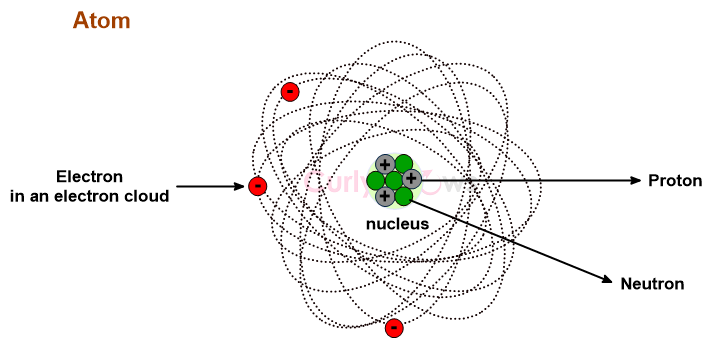
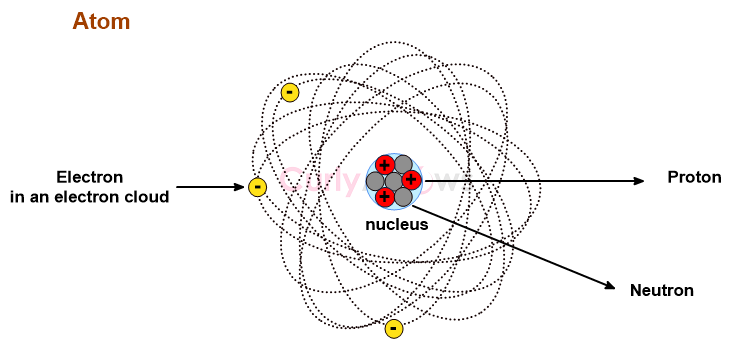
Commonly, it is accepted that a compound is an Arrhenius acid if it liberates hydrogen ions as H+ in water. In the next step, these H+ ions combine with water molecules to form hydronium ions (H3O+). The two steps can be summarised to say that Arrhenius acids are compounds that form hydronium ions, provided water is the solvent.