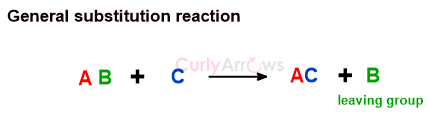
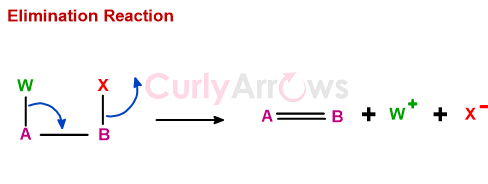
Zaitsev’s Rule
A rule stating that an elimination reaction will give as the major product the most stable alkene, that is, the alkene with the most highly substituted double bond.
For example, dehydrohalogenation of alkyl halide 2-Bromo-2-methylbutane can proceed in two ways to give two types of alkenes, of which only one is a major product.