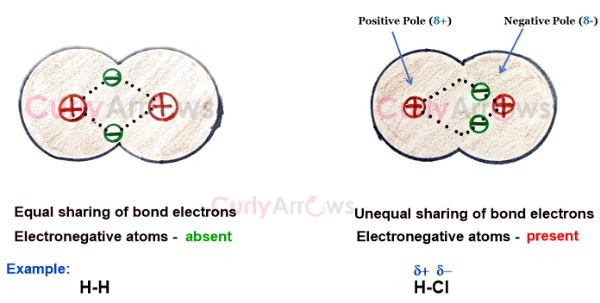
How can an electronegativity scale predict the type of bonding ?
Following the footsteps of Gilbert N. Lewis in understanding the nature of the covalent bond, Linus N. Pauling stumbled upon an interesting observation. Pauling noticed that the nature of the chemical bond could be explained using a scale or continuum.