Common Reactions involving Carbon - Centered Free Radicals
Learning Objective: To learn about a few common reactions that carbon-centered free radicals undergo in organic chemistry.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To learn about a few common reactions that carbon-centered free radicals undergo in organic chemistry.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To learn the two different ways a free radical reacts and the mechanism it undergoes.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To learn how to predict the stability of a hydrocarbon to form a free radical using the dissociation energy value of the C-H bond.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To study the structural features that contribute to the stability of a carbon-centered free radical in organic chemistry.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To study the structural features of a carbon-centered free radical in organic chemistry.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To learn about different ways by which the formation of a carbon-centred free radical occurs.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To learn about various methods by which a carbanion reacts with other positive centers in organic chemical reactions.
Skill Level - Advanced
Prerequisites:
Learning Objective: To compare different molecular structures with their pKa values to understand which structures can easily form and effectively stabilize a carbanion.
Skill Level - Advanced
Prerequisites:
Acidity
Learning Objective: To learn different methods to stabilize a carbanion.
Skill Level - Advanced
Prerequisites:
Learning Objective: To learn about the three ways carbanion intermediates are generated.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To learn about the structure, type, and nature of the carbanion intermediate part of organic chemistry reactions.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To understand the four common organic reactions that generate carbocation intermediates.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To study the fate of a carbocation intermediate in a chemical reaction.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To learn about the structural feature that stabilizes also destabilizes the carbocation intermediate.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To study the two methods of how carbocation forms.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To study carbocation as the reactive intermediate - formation, structure, and types.
Skill Level - Intermediate
Prerequisites:
Learning Objective: To study what reactive intermediates are, their types, and their role in organic chemistry.
Skill Level - Intermediate
Prerequisites:
Benzyne | Benzyl | |
|---|---|---|
Identification | Benzyne is identified as a triple bond between two adjacent carbon atoms in a benzene ring. Root- Benzyne (like the triple bond of alkyne) |
Benzene | Benzyne | |
|---|---|---|
Identification | Alternating single and double bonds. | Alternating single and double bonds plus an extra pie bond (triple bond) between two adjacent carbons. |
Structure |
Radical initiators are chemical species that quickly produce free radicals by light and heat exposure. Examples include Halogens (Cl2), AIBN (R-N=N-R), peroxides (R-O-O-R), acetophenone, benzophenone, etc.
A radical reaction usually happens in three steps- Initiation (creation of free radicals), Propagation (multiplication and growth of radical reaction), and Termination (stopping of the free radical reaction).
Radical inhibitors are chemical species that inhibit radical reactions by halting the chain propagation step.
Examples of such inhibitors are Hydroquinone, BHT (2,6-di-t-butyl-4-methylphenol), etc.
Free Radicals | Carbocations |
|---|---|
Free radicals are electron-deficient atoms or groups of atoms. It can include carbon-centered free radicals (for example, H3C.) and non-carbon-centered free radicals (for example, HO., Cl., etc.). | Carbocations are electron-deficient carbon atoms. |
Free radicals do not carry any charge and are neutral species. |
Free radicals are generated due to the homolytic bond cleavage, a type of bond breaking where each atom holding the two-electron covalent bond gets one electron. Such a homolytic bond cleavage requires an input of energy, either in the form of heat or light.
Learning Objective: To study the history, nature, examples, and types of free radicals, a reactive intermediate in organic chemistry.
Skill Level - Intermediate
Prerequisites:
A carbocation is a positively charged, electron-deficient ion of carbon with only 6 valence electrons from the preferred 8 for covalent bonding. Therefore, it acts as an electron-accepting reactive intermediate in many organic reactions.
After accepting two electrons, it can transform from a less stable to a more stable uncharged molecule.
A carbocation is a positively charged, electron-deficient carbon ion that acts as a reactive intermediate in many organic reactions.
The central carbon in a carbocation has only 6 valence electrons instead of the preferred 8 by covalent bonding. As a result, it has only three covalent bonds and, therefore, trivalent.
A carbocation is a reactive intermediate with only 6 valence electrons from the usual 8 electrons for carbon by covalent bonding. This makes the carbon atom bear a positive charge and possess three bonds instead of four.
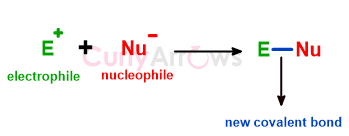
Carbocations and electrophiles are both electron-deficient species and therefore are electron acceptors. They are attracted to electron-rich centers. However, the electrophile is slightly different from carbocation.
Electrophiles can have a full positive or partial positive charge due to electronegative atoms creating polarity differences, or they can be neutral uncharged species with vacant orbitals.
For example-
A carbocation is a positively charged, electron-deficient carbon atom that acts as a reactive intermediate in many organic reactions.

A carbocation is a positively charged, trivalent carbon ion that acts as a reactive intermediate in many organic reactions.
With three bonds and only six electrons, carbocations have an incomplete octet and, therefore, electron deficient. It functions as an electron acceptor and an electrophile forming new Carbon-Carbon (C-C) bonds.
A carbanion is a negatively charged, trivalent carbon ion that acts as a reactive intermediate in many organic reactions.
With three bonds and 8 electrons, carbanions are electron rich and act as a base forming a C-H bond or as a nucleophile forming a C-C bond.
Electrophiles are electron-deficient species that accept electrons from other electron-rich counterparts, the nucleophiles, to form a two-electron covalent bond.