Intermolecular forces are collective forces in organic chemistry that cause atoms and molecules to stick together and interact; therefore, they are electrostatic in nature. The collective strength of these electrostatic interactions can affect a compound's physical properties, like melting and boiling points, density, refractive index, solubility, and others.
Almost all atoms and molecules interact through intermolecular forces, an inherent property resulting from the presence of electrons.
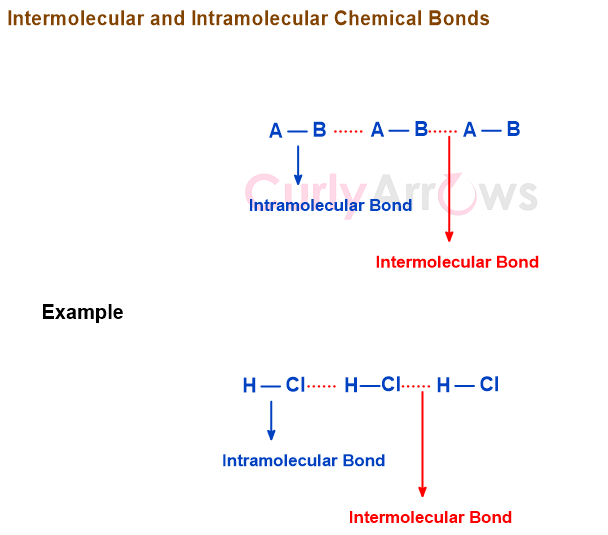
Intermolecular forces differ from intramolecular forces since the latter occur within the same molecule. Intermolecular forces require separate atoms or molecules to be within at least 0.1-0.5 nm of each other. Therefore, they operate at short distances, with their strength waning with increasing distance.
The presence of an ion increases the strength of the attractive force, increasing the interactions. The strength of intermolecular interactions also depends on the molecule's polarity, with polar-polar interactions being stronger than polar-nonpolar interactions and nonpolar-nonpolar interactions being the weakest.
The most potent intermolecular forces in organic chemistry are ion-dipole, ion-induced dipole, van der Waals (Debye, Keesom, London), and Hydrogen bonding interactions.
Intermolecular forces are considered nonbonding interactions in organic chemistry.
