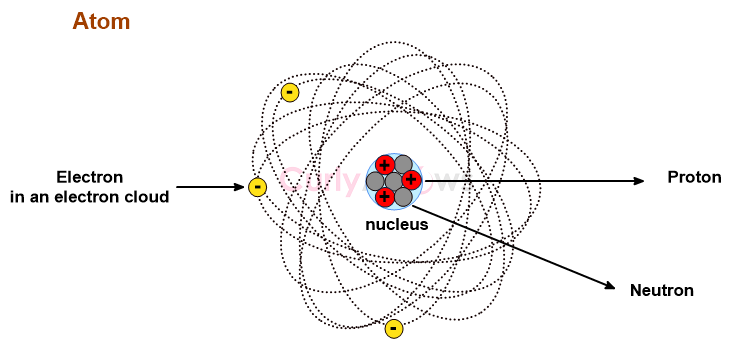
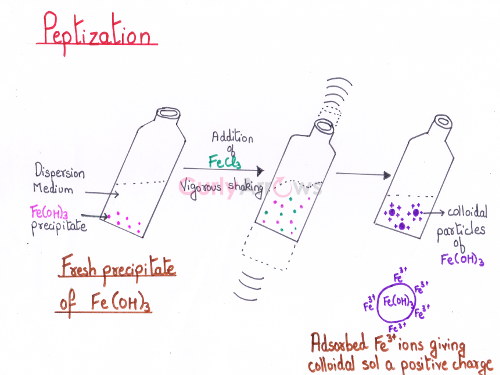
Periods
Periods in chemistry refer to the horizontal rows of elements in the periodic table, which represent all 118 elements across 7 rows.
The periods run from left to right, with each consecutive element increasing in atomic number by 1.
Periods 1, 2, and 3 have 2, 8, and 8 elements, whereas periods 4, 5, 6, and 7 have 18, 18, 32, and 32 elements.