A proton is one of the subatomic particles found in every atom’s nucleus, denoted as p or p+.
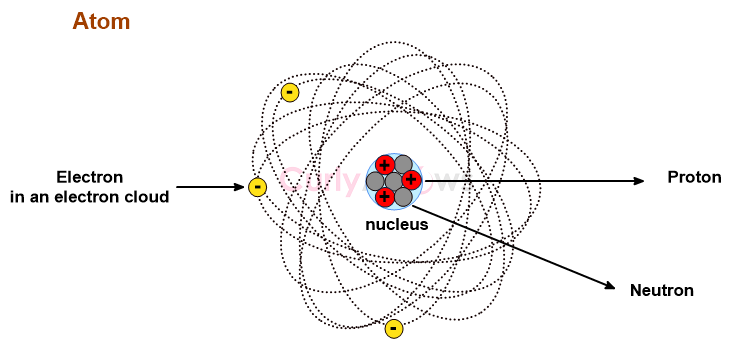
A proton has a positive charge of +1 and a mass of 1 AMU.
1 AMU (atomic mass unit) equals 1.6726216 X 10-27 kg.
The number of protons in the nucleus of an atom is fixed and, therefore, helps to identify an element. Changing the number of protons is a nuclear level transformation that changes an atom's identity and forms a new element.
Therefore, the proton number equals the atomic number (Z). For example, an Oxygen atom with eight protons in its nucleus has an atomic number 8.
All the 118 elements of the modern periodic table are arranged in order of their increasing number of protons.
The hydrogen ion H+ is also referred to as a proton in several organic chemistry reactions because the loss of the valence electron from a neutral hydrogen atom leaves only the hydrogen nucleus—a proton.

