A bond angle is a geometrical angle between two bonds originating from the same central atom in a covalently bonded molecule, measured in degrees (o).
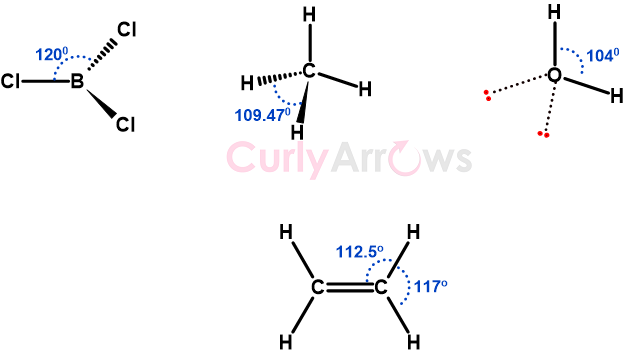
In other words, it is the angle between three atoms of which one is a central atom, and they are all held by covalent bonds.
The formation of the bond angle between atoms is mainly due to the electrons, both bonding and nonbonding, a concept best explained by the VSEPR theory.
According to VSEPR theory, once a central atom undergoes hybridization and subsequent bond formation, the bonding and the nonbonding electrons arrange themselves to avoid electron-electron repulsions. This is responsible for forming different bond angles and creating various molecular shapes.
The maximum and minimum distance between a set of electrons ranges from 180o to less than 90o. The bond angle of 72o is also seen in polyatomic molecules having considerable strain, for instance- IF7, etc.
Bond angles, therefore, are responsible for organic molecules having three-dimensional shapes; the most common ones are- linear, trigonal planar, tetrahedral, trigonal planar, and octahedral.
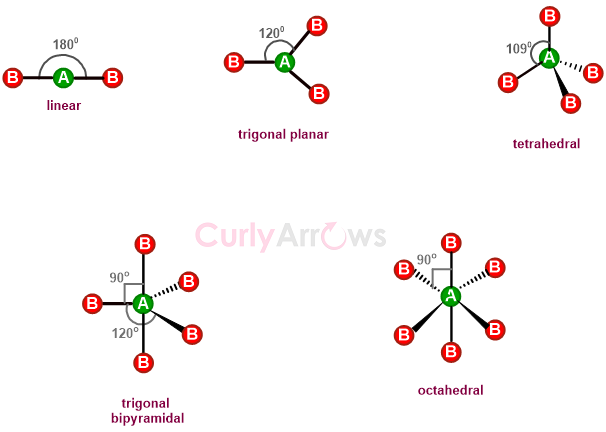
Though the nonbonding electrons (or lone pairs) in the central atom are not used in calculating bond angles; however, their presence creates repulsive interactions with the bonding electrons (of the bonds).
So, this brings a further change in the bond angles of the basic shapes to create more varied shapes like bent, T-shaped, seesaw, trigonal pyramidal, etc.
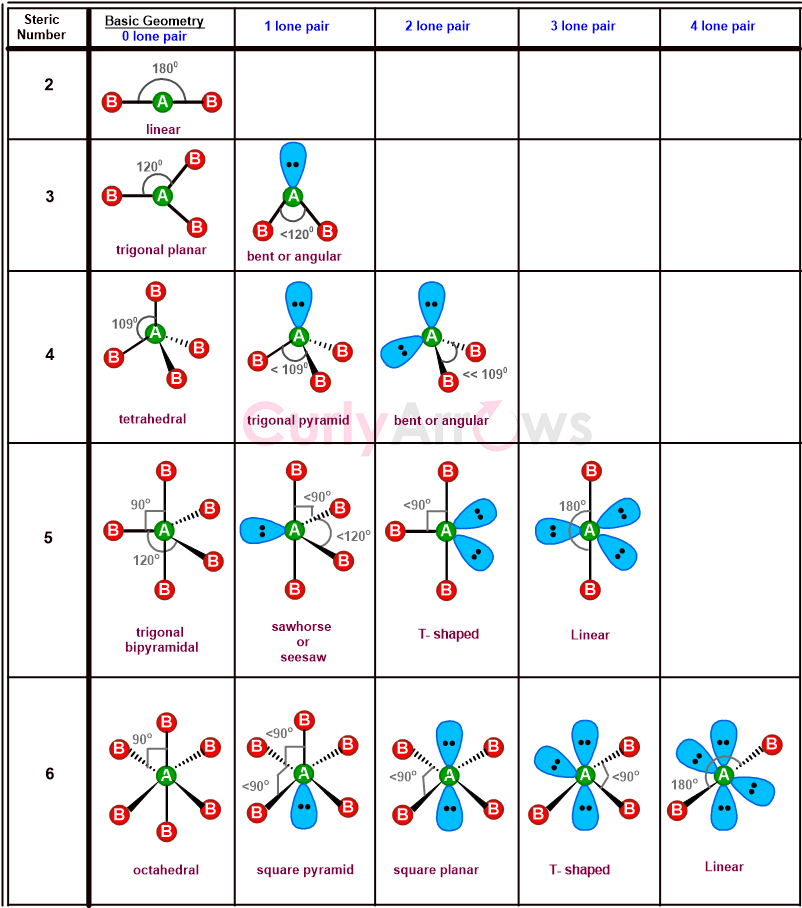
The shapes, in turn, influence various molecular properties like boiling point, reactivity, color, polarity, biological activity, etc.
Related Comparison - Differences between Dihedral Angle and Bond Angle
