The melting point of a substance is the temperature at which a solid, ordered crystalline substance is in equilibrium with a more random liquid state, thereby undergoing a phase change.
Since the components of the solid substances are bound by strong intermolecular attractive forces, it also reflects the energy needed to weaken these attractive forces and move freely in the liquid state.

An ice cube is a good example to understand the concept of the melting point. At temperatures below 0 °C and at 1 atm, water molecules condense to form solid, crystalline ice.
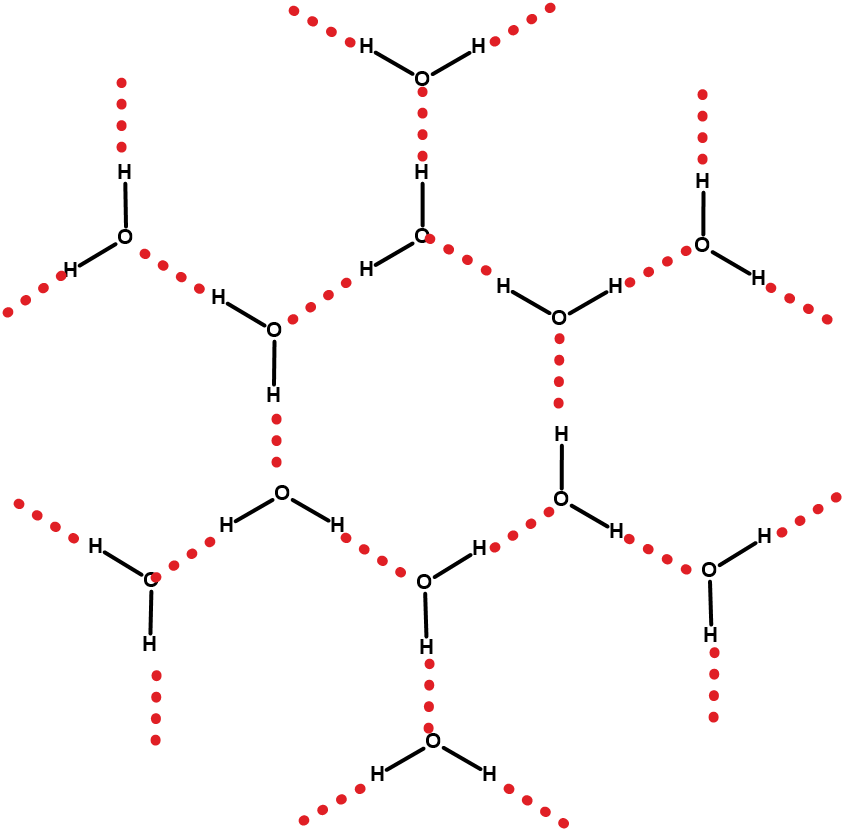
However, as the temperature rises to 0 °C, the molecules of water (or H2O) acquire heat (or thermal) energy required to vibrate and eventually overcome the attractive forces. The molecules separate and start to move freely. The temperature at which the ice begins to undergo a phase change into water is recorded as the melting point of ice.
At the melting point, there exists a dynamic equilibrium between the solid and the liquid state.
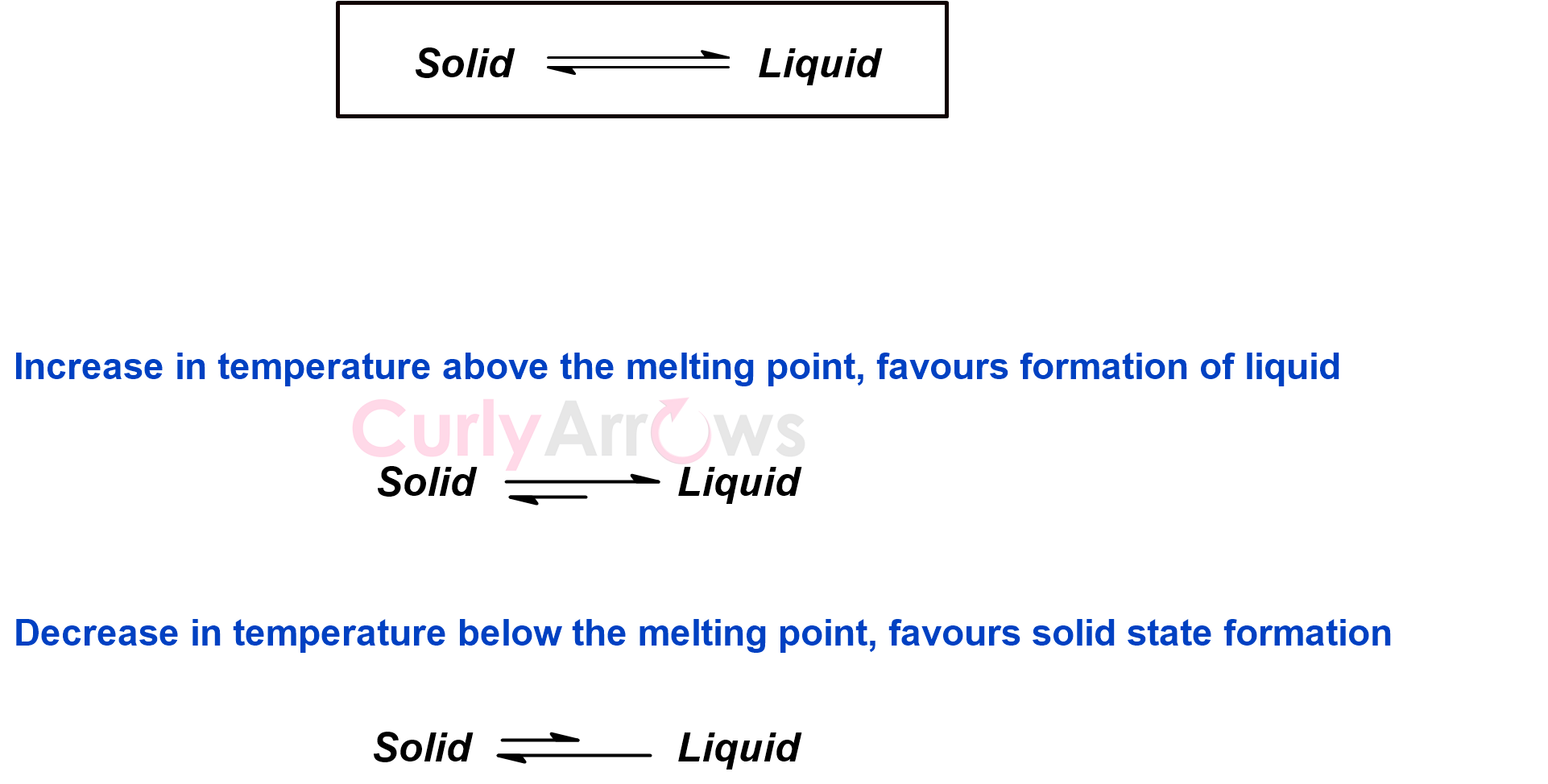
Different substances have different melting points. Crystalline solids have a sharp melting point, whereas amorphous solids melt at a range of temperatures. Therefore, the melting point becomes a crucial physical property for identifying substances.
The factors affecting the melting point include intermolecular forces, molecular structure and packing, molecular size and weight, purity of the substance, pressure, and polarity.
