The activation energy (Ea) is the minimum amount of extra energy absorbed by the reactant molecules from an energy source such as heat, light, etc., to attain the threshold value for effective collisions to occur and thereby to form an intermediate complex (active state) that finally results in product formation.
In principle and visually, you can see below that it is the energy barrier that must be overcome for a chemical reaction to occur, that is, for the reactants to go from the initial to the product stage via an intermediate activated complex formation.
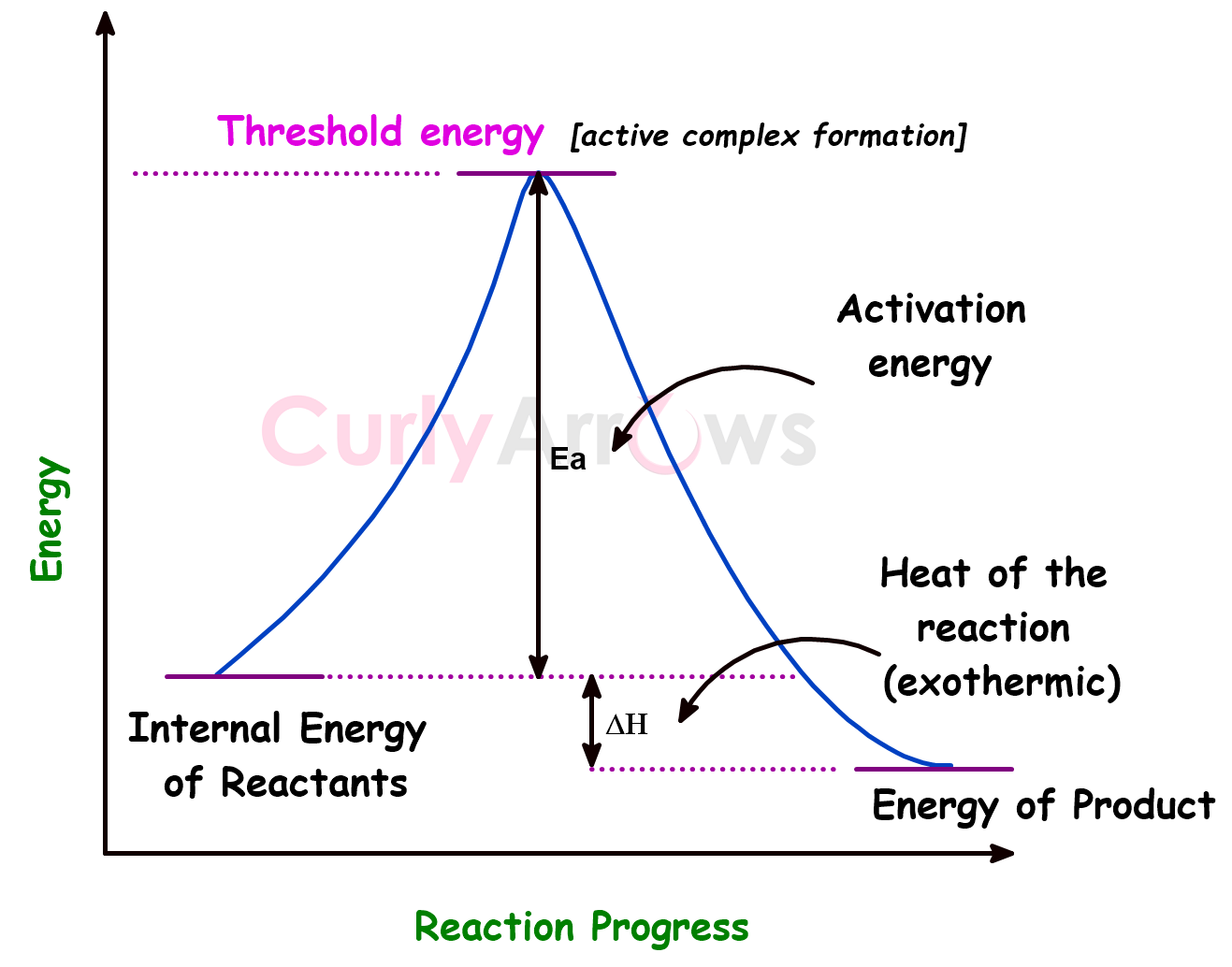
Only collisions (since kinetic theory considers reactants as hard spheres undergoing collisions) with energy ≥ Ea can lead to a reaction.
It can also be expressed as the difference between the threshold energy and the average kinetic energy possessed by the reactant molecules at the start of a chemical reaction.
Therefore,
Activation Energy = Threshold Energy – Average Kinetic Energy of the reactant molecules
In short, the activation energy is the energy hill that reactants must climb to become products.
A lower hill (obtained using a catalyst) or giving the reactant molecules extra energy in the form of heat or light to climb the hill can lead to a faster reaction.
