As a reactive species, a methylene refers to an electron-deficient carbene, represented as :CH2.
Carbenes are carbon-containing electrophiles that contain only 6 valence electrons instead of the standard eight required for stability. It has an unshared electron pair and two bonds, making it a divalent species, highly unstable, having a fleeting existence. Though classified as electrophiles, methylene species have no formal charge and are neutral.
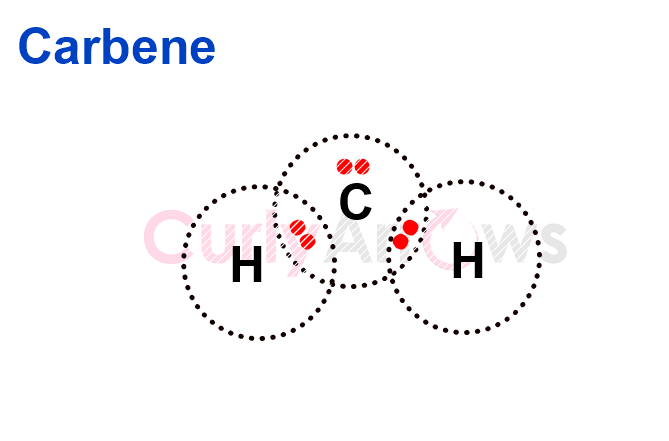
As a group, methylene refers to the -CH2- unit of a carbon chain in organic compounds. It is a divalent group and forms part of the carbon backbone of hydrocarbons. It can also be found next to various functional groups, as part of the central carbon chain.
For example, a methylene group occupies a central position when part of hydrocarbon propane (CH3-CH2-CH3) or when the methylene group is attached to any functional group, like in the case of propanal (CH3-CH2-CHO).
It is part of a repeating unit of a polymer chain, in the case of polyethene.
Repeating methylene units can add nonpolarity in molecules.

