The different spatial arrangements organic molecules adopt due to the rotation of the single bond are called conformations. A specific conformation is called a conformer or conformational isomer.
Many such conformational isomers are undoubtedly interconvertible by single bond rotations. So, these isomers are simply different rotational (or structural) arrangements of the same molecule.
It is mostly hard to isolate pure conformers because the molecules are rapidly rotating and adopting infinite possible conformations. However, the most common conformations are those where, by adopting a particular arrangement, the potential energy of the molecule is minimum, which hints at its higher stability.
In such molecules, the energy required to undergo bond rotations (called the rotational energy) is higher, implying a higher barrier for bond rotations.
Conformational isomers are a type of stereoisomers. However, they are distinct from configurational isomers.
Conformations can be formed by open-chain (acyclic) or closed-chain (cyclic) carbon compounds. Examples of some common conformations adopted by carbon-carbon single bonds containing organic molecules are- eclipsed, staggered, gauche, planar, boat, and chair conformations.
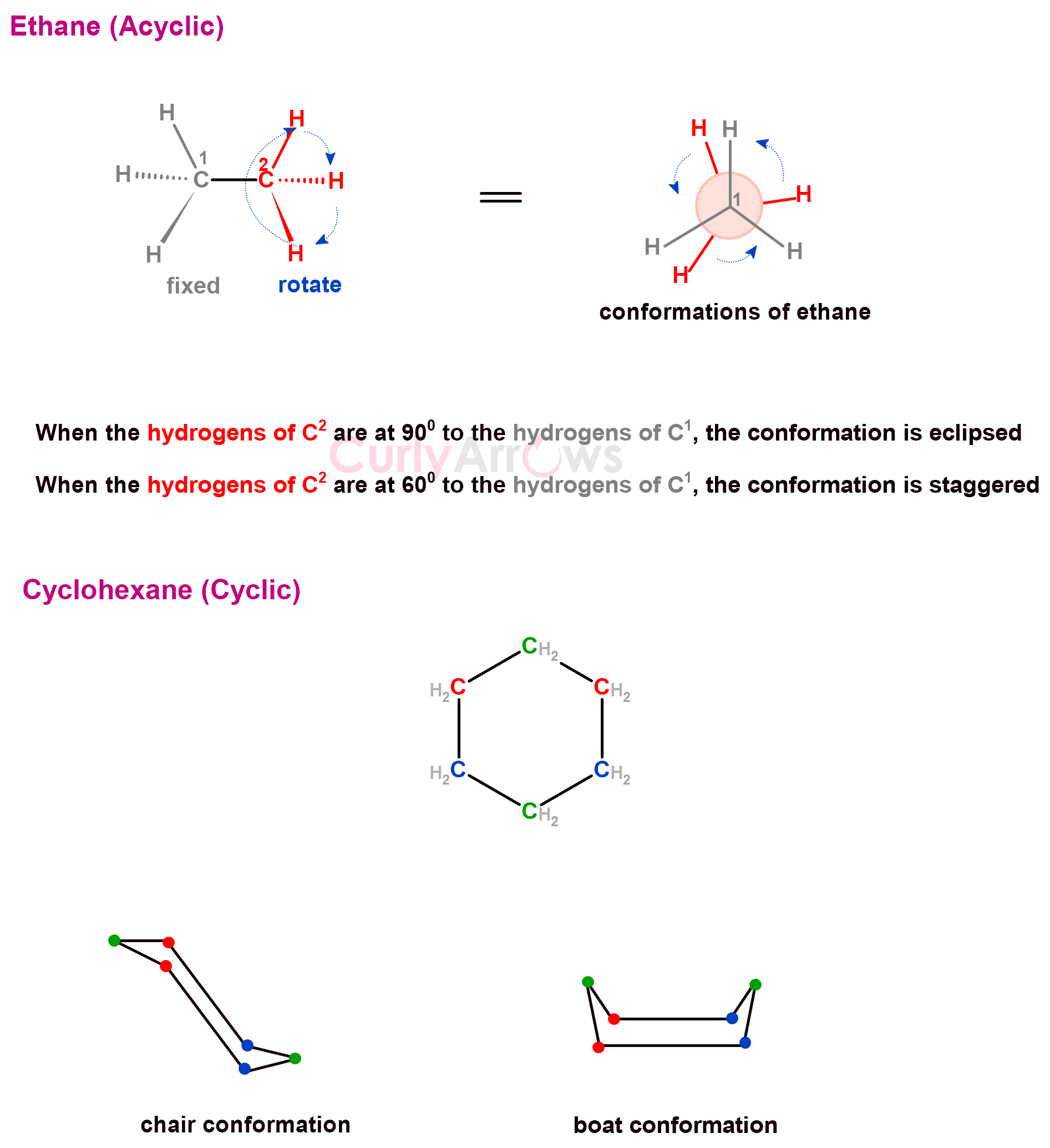
Some common terminologies associated with conformational isomerism are – eclipsed (or syn), staggered (or anti), gauche, axial, equatorial, Newmann projections, torsional strain, Rotational energies, boat conformation, chair conformation, and ring-flip.
Related Foundational Chapter: Covalent Bond (Covalent Bond Properties- Rotation and Breakage) (subscribers only premium content)
