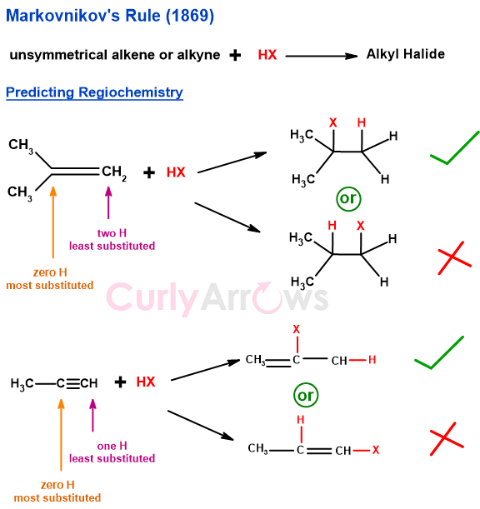
Russian chemist Vladimir Markovnikov proposed Markovnikov’s rule in 1869 to predict the regiochemistry of addition reactions between unsymmetrical alkenes/ alkynes and hydrogen halides to form alkyl halides.
According to the rule, ‘An alkyl halide is formed in a reaction between an unsymmetrical alkene or alkyne and hydrogen halide (HX) when the negative part of the reagent (X-) attaches to the alkene carbon that has fewer number of hydrogen atoms across the double bond.’
The alkene carbon with fewer hydrogen atoms is the one that is the most substituted with alkyl groups.
Modern expression-
In the ionic addition of an unsymmetrical reagent to a multiple bond, the positive portion of the reagent (the electrophile, in the example above H+) attaches itself to a carbon atom of the reagent in the way that leads to the formation of the more stable intermediate carbocation.
Read more in detail about the Markovnikov’s Rule.
