Learning Objective: To learn about the strongest chemical bond in chemistry, the ionic bond.
Skill Level – Intermediate
Prerequisites:
Related –
Chapter: Bonding in Atoms
Author's Note: The study of organic chemistry sometimes involves ionic compounds, which play a key role in many reactions, such as SN1/SN2, acid-base reactions, organometallic additions, and phase-transfer catalysis. We will also see ionic character extensively when we talk about the salt of organic acids (e.g., sodium acetate, CH₃COONa — ionic between Na⁺ and the acetate anion, but the anion itself is covalent), ionic liquids, organometallics like RMgX, and zwitter ions like ammonium salts. In addition, ionic compounds form strong chemical bonds; therefore, their study is imperative for understanding reactions, solubility, acidity, and catalysis.
In this section, we will learn what an ionic bond is and how strong it is. We will then see how they form and hold in various lattices, what their characteristics are, and how they differ from covalent bonds.
Introduction to Ionic Bonding
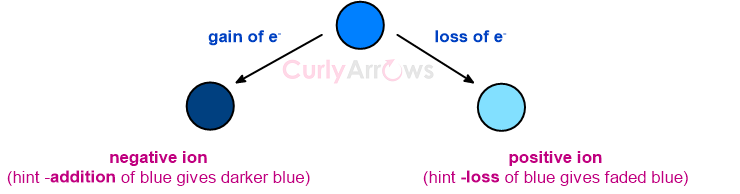
A charge-containing atom is called an ion. The charges are formed when an atom gains or loses electrons. On gaining electrons, the extra electrons are shown as a negative charge (like in Cl-), and the loss is expressed with a positive charge on the atom (like in Na+).
The electrostatic attraction of the oppositely charged ions (like Na+ and Cl-) creates a chemical bond known as the ionic bond, also known as an electrovalent bond.
In other words, ions form an ionic bond.
Ionic bond is very strong electrostatic attraction between oppositely charged ions and requires between 400-4000 kJ/mole of energy to separate them into gaseous atoms.
To understand the mightiness of the force involved, imagine a regular table salt that is so stable and unreactive at normal room temperature. The salt is Sodium Chloride (NaCl), composed of oppositely charged ions, Na+ and Cl-, held firmly in a lattice by the strength of the ionic bond. The solid NaCl requires a very high temperature of 800°C to melt. The caged lattices that hold the ions in place are only broken at the melting point of 800oC. To put things in perspective, a pot of water or sunflower oil boils at 100oC and 227oC, and salt melts at a much, much higher temperature.
To further separate the ions into parent elements, sodium metal, and Cl2 gas requires even more energy, like electrical energy to be passed through the molten solution. Such a strong bond between ions Na+ and Cl- is the best example of the strength of an ionic bond.
Ionic Bond Formation
An ionic bond is one of the two ways (the other is a covalent bond) by which atoms attain a stable noble gas configuration.
Read - The Octet Rule
In an ionic bond, the atoms attain noble gas configurations by transferring electrons from one atom to another: one a metal and the other a nonmetal.
If an atom (a metal) loses an electron, it has more protons in its nucleus than electrons in its shell, causing an imbalance. The equal ratio of the atom's positive and negative charges is thereby disturbed.
The atom becomes a positively charged ion and is called a cation (mnemonic - the t in cation looks slightly like a + sign indicating a positively charged ion). The number of electrons lost is written in the superscript with the corresponding positive charge. Example, Na+, Mg2+ etc.
Elements of Group 1, 2 and 3 would lose one, two and three electrons and become a cation with a +1, +2 and +3 charge.
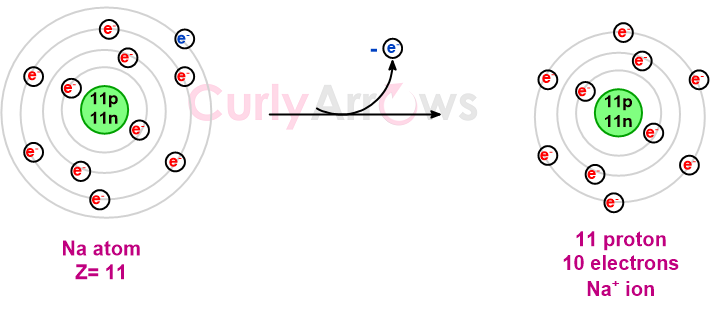
The electron lost by the cation is picked up by the other nonmetal atom that is willing to accommodate the extra electrons in its shell.
Post the uptake, the nonmetal atom has more electrons in its shell than protons in its nucleus. Once again, there is an imbalance between the positive and the negative charges, with extra electrons than the nuclear protons. Such an atom is denoted with a negative charge and is called an anion (mnemonic - anion has an n for a negative charge). This number of electrons an atom accepts is written in the superscript with the corresponding negative charge - for example, Cl-, O2- etc.
Elements of Group 17, 16, and 15 would gain one, two and three electrons and become an anion with a -1, -2, and -3 charge.

The anions and the cations are stable due to attaining their corresponding inert gas electron configuration.
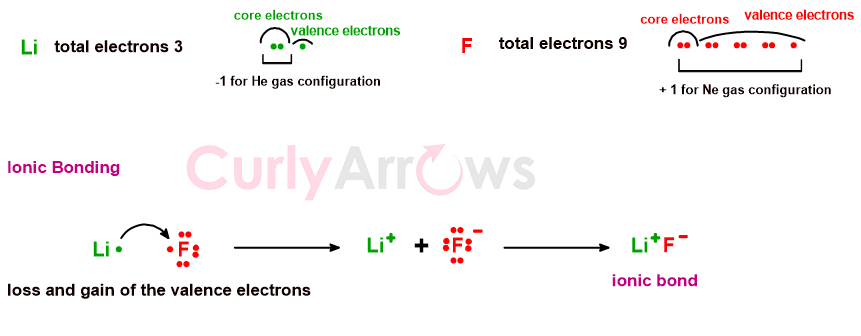
For example, lithium has an electron in excess from the nearest Helium gas configuration, and Fluorine has one electron less than the Neon configuration.
.png)
Lithium can easily lose its valence electron, Fluorine can gain one, and the transfer of one electron gives both a noble gas configuration. The ions formed can electrostatically interact and create the ionic compound Lithium Fluoride (LiF) by an ionic bond.
Another example is of ionic compound MgCl2. Mg loses two electrons to reach a stable Neon gas configuration. The Chlorine atoms pick the two electrons to attain the Argon gas configuration.
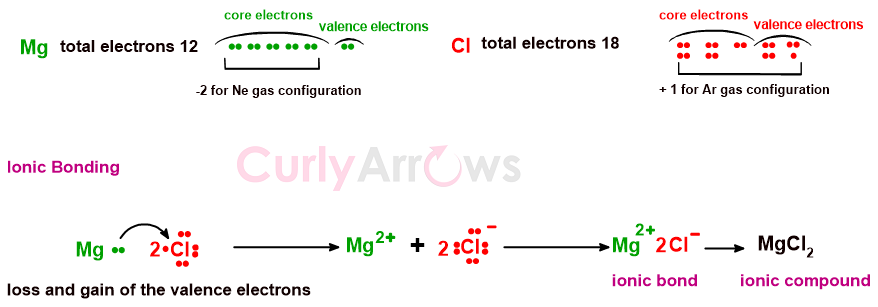
Once the negatively charged ion balances the positively charged ion such that the number of electrons lost and gained remains in correct stoichiometric quantities, an ionic bond is formed. Once combined, the ions are arranged in strong lattices releasing a large amount of energy, known as the lattice energy.
Characteristics of Ionic Bonding
Ionic bonding phenomena form a large crystal lattice, a regular, repeating arrangement of the ions in a 3-dimensional space, rather than forming individual molecules like covalent bonding. The positive ions are tightly packed with the negative ions in various cubic and hexagonal lattice arrangements in the solid state.
According to its size or radius, a cation can attract multiple anions and vice versa to create a regular, continuous geometric lattice pattern. Stronger ionic bonds result in stronger crystalline solid lattices reflecting the ionic compound's higher stability. The ionic compound's crystal lattice stability is measured in lattice enthalpy values. The greater value corresponds to a stronger, tighter lattice.
Therefore, ionic bonding is common in inorganic compounds but relatively uncommon in organic compounds.
Author’s Note - In total, there are theoretically 14 Bravais lattices possible in crystalline compounds in various categories of ionic, covalent molecular, metallic, covalent network, etc., but ionic compounds predominantly adopt cubic-based ones (simple cubic- like in CsCl, FCC- like for NaCl, BCC) due to homogenous, non-directional (isotropic) electrostatic forces and ion packing rules.
Subscribe to the study program on Organic Chemistry Fundamentals.
Next: Formation of Ionic Compound
Next: Requirements for Ionic Bonding
