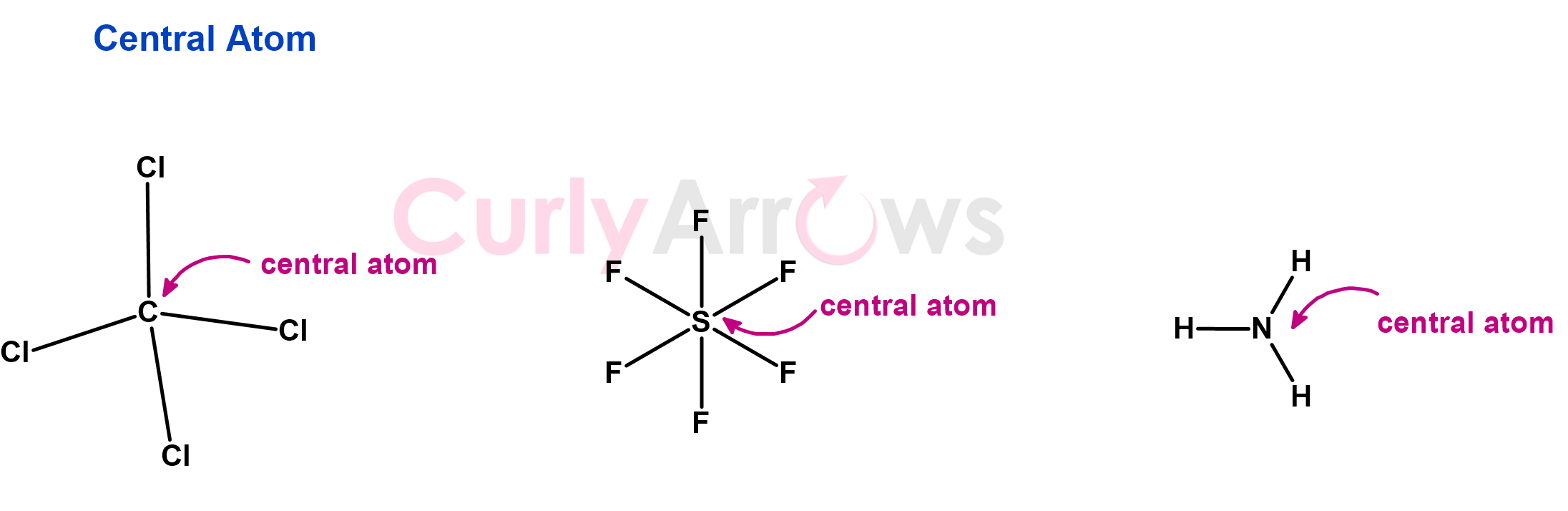
In molecules consisting of more than two atoms, the least electronegative atom (except Hydrogen) is the central atom. Due to its low electronegativity, the central atom will not hoard electrons but will share with other atoms, thereby forming a maximum number of bonds than the terminal atoms. So, the central atom is also the least numerous.
It is written first in the molecular formula and assigned the lowest subscript. For example, the carbon in CCl4 is the central atom and is the least electronegative compared to Chlorine. Other examples are S in SF6, N in NH3, C in C2H5OH, etc.