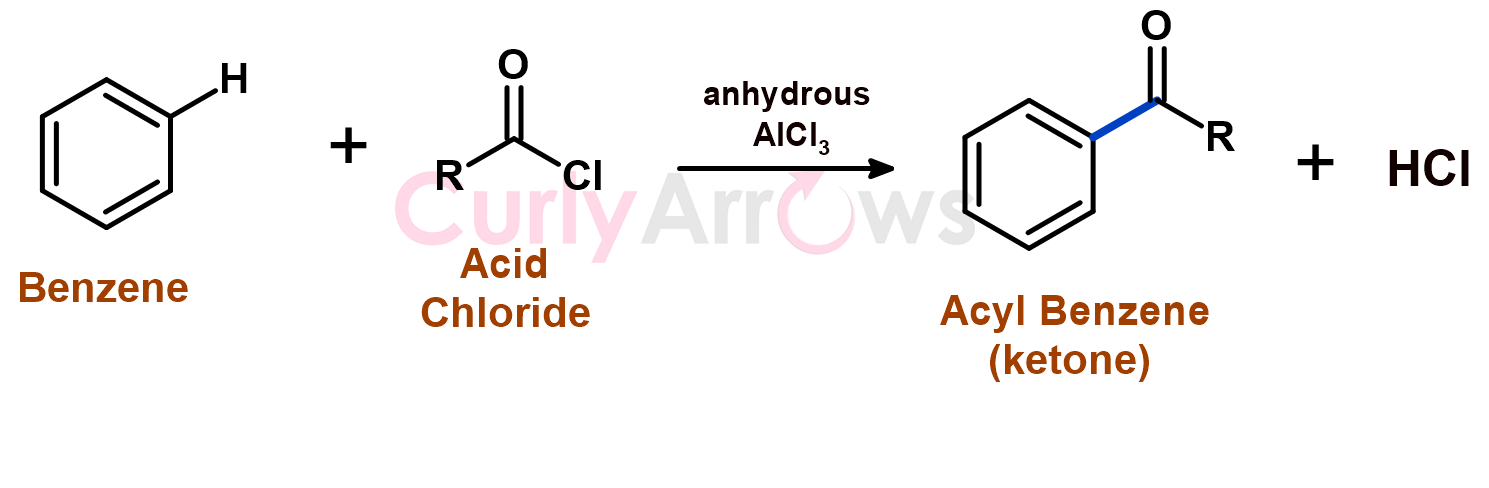
Friedel Craft acylation reaction is a type of electrophilic aromatic substitution reaction wherein the hydrogen of the aromatic ring is substituted with the acyl group (R-CO-), and a new Carbon-Carbon bond is formed. Such an acylation reaction requires a Lewis acid catalyst such as AlCl3 and an acid chloride (R-CO-Cl). The byproduct formed is HCl.
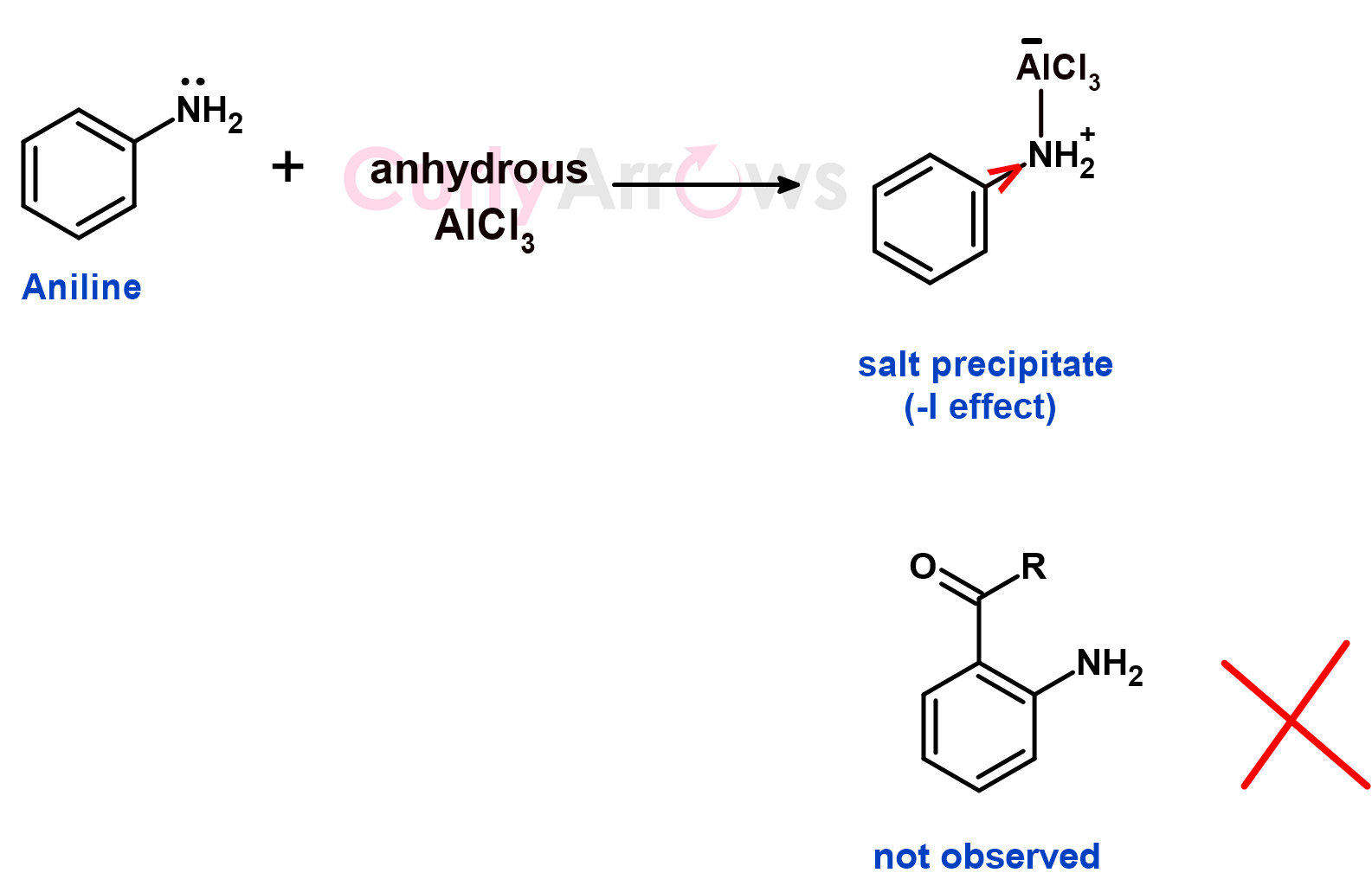
Under the Friedel Craft Reaction condition, the aniline (Lewis base) binds to the electrophile AlCl3 (Lewis acid) to form a coordination complex (salt).
Acid + Base = Salt
The positive charge on the nitrogen is electron withdrawing, and it pulls the electron density of the ring by negative inductive effect (-I effect). It, therefore, deactivates the ring for further acylation reactions. The complex precipitates out of the reaction mixture, and the acylation reaction is not observed.
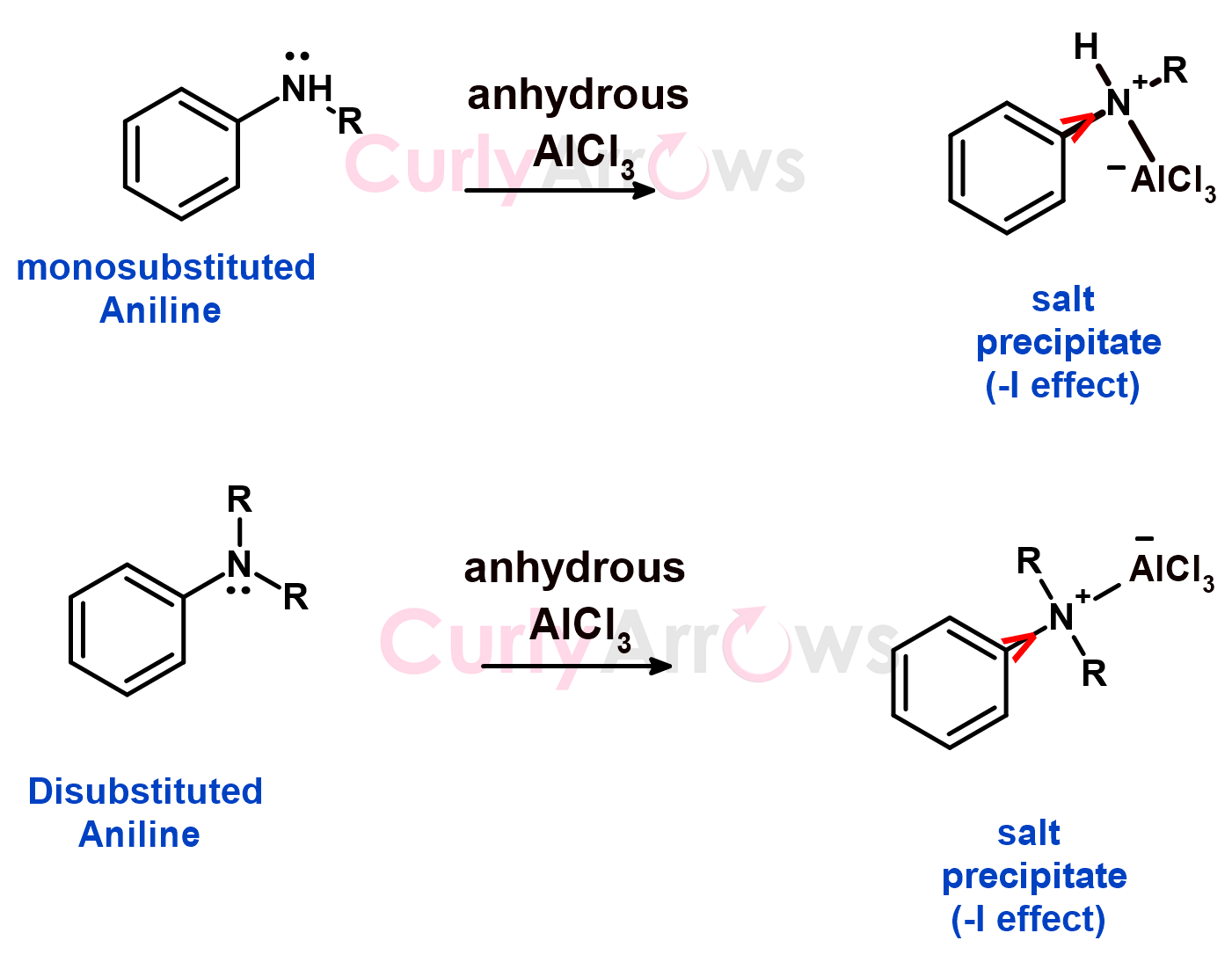
A similar case is seen for mono and disubstituted anilines.
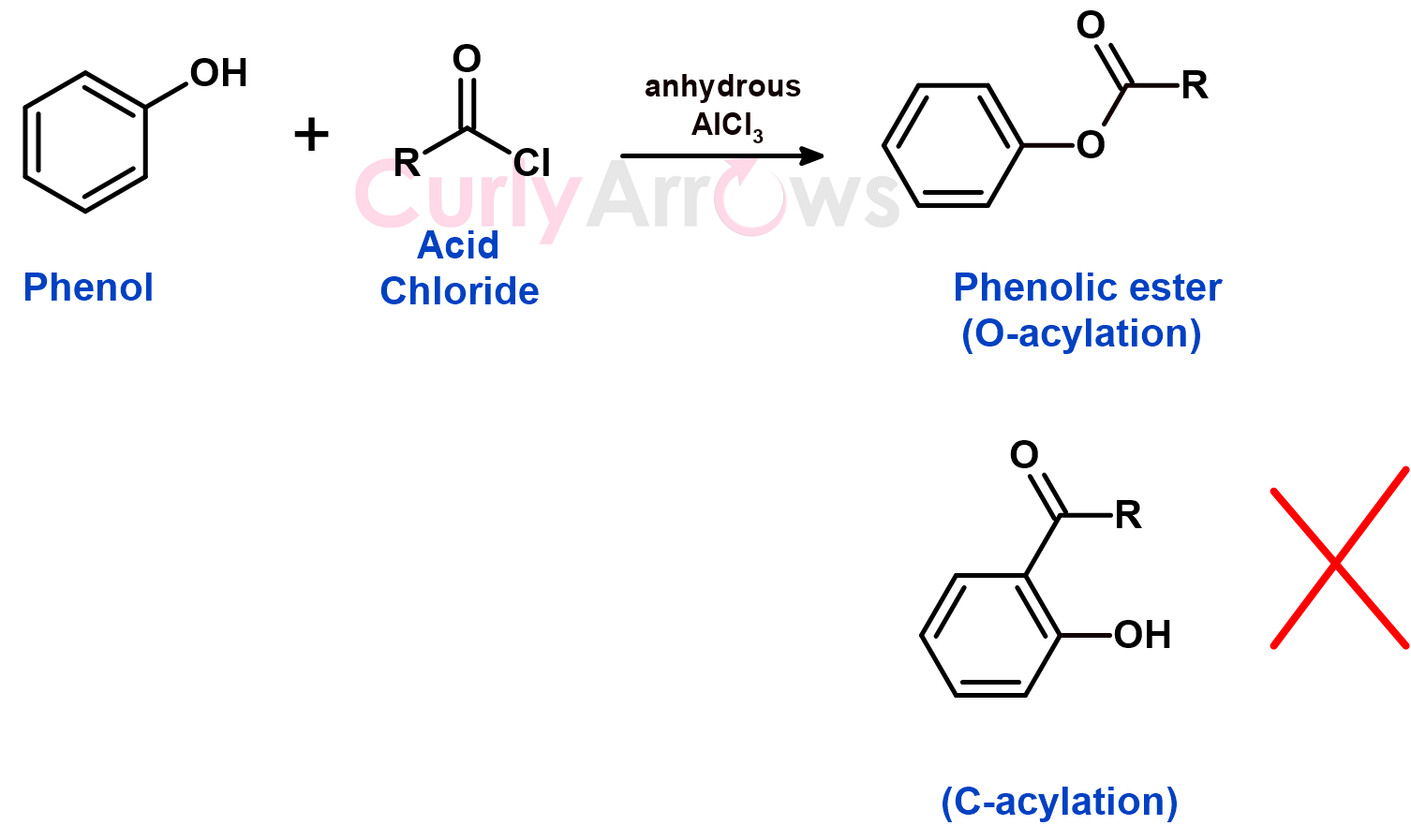
In the case of the Phenols, the acylation takes place at the oxygen (O-acylation) and not at the carbon of the ring (C-acylation) to form a phenolic ester.
Nevertheless, a method does exist to convert the O-acylated product to the C-acylated product. The O-acylated product can be treated with an excess of AlCl3 (Fries rearrangement) to obtain ortho and para hydroxy acetophenone. The ortho and the para isomer can be later separated by chromatographic techniques.
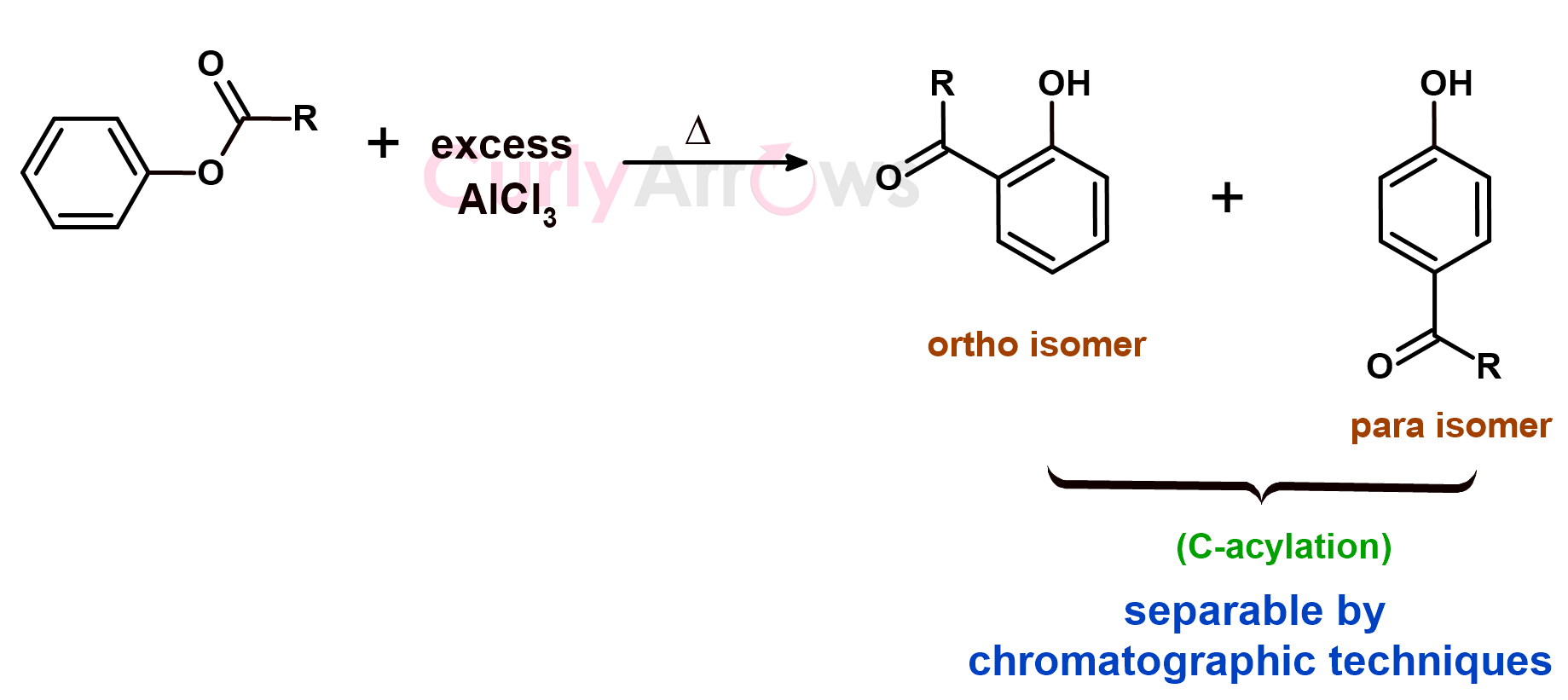
The Phenol does not form a coordination complex with AlCl3 similar to the aniline as the lone pair on the -OH is tightly held and is less basic than the -NH2 group.
But if the oxygen of the phenol is substituted, (example, Anisole) it then prefers to undergoes acylation reaction at the carbon of the benzene ring (C-acylation). The para isomer would be the major product.
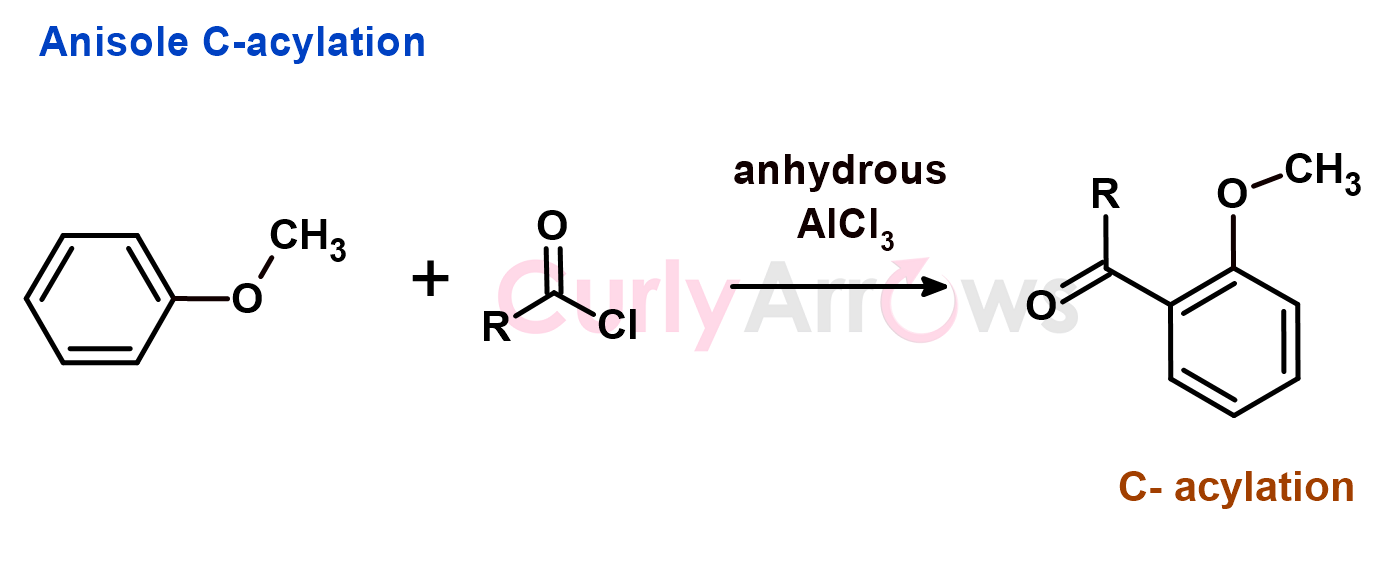
In summary, Phenols do not undergo FC acylation reaction because of O-acylation and in the case of Anilines, it forms a salt with AlCl3 and precipitates out of the reaction mixture.
Suggested Reading- Friedel Craft Acylation Reaction
