Learning Objective: To study with examples the hydrogen bond donors and acceptors.
Skill Level – Intermediate
Prerequisites:
Chapter: Intermolecular Forces
Sub-topic: Hydrogen Bonding
Author's Note: Interaction is a two-way process; one gives and the other accepts. Hydrogen bonding interaction follows a similar protocol, it requires a Hydrogen bond donor and a Hydrogen bond acceptor atom. Still, let the name not fool you, in the context of hydrogen bonding, there is no actual bond formation that occurs here. This section covers what type of atoms to look out for when speaking about Hydrogen bonding. The table covers molecules that perfectly satisfy the donor and acceptor atom requirements and those that don't.
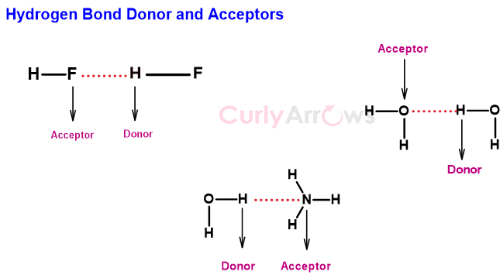
The donor and acceptors atoms in the Hydrogen bond
The Hydrogen covalently bonded to an electronegative atom is called the donor atom. The donor atom is always found in between the two electronegative atoms.
The acceptor atom is the other electronegative atom that electron starved Hydrogen tries to attract.
Though the name is Hydrogen bond donor, the molecule does not donate the Hydrogen as protons but barters its positive charge for the electron density of the neighboring electronegative atom. At the same time, it maintains its covalent bond character.
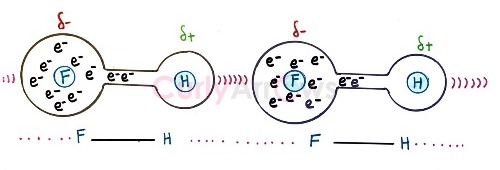
Therefore, the donor must have at least one Hydrogen atom in their molecule to form a Hydrogen bond. Examples of Hydrogen bond donor molecules are NH3, H2O, HF, ROH, NH(R2), and NH2R.
The Hydrogen bond acceptors are also electronegative atoms -N, O, or F possessing a minimum of one lone pair electron, which it can contribute to the electron deficient Hydrogen of the donor atom.
Lone pair is a set of two outermost valence electrons of an atom that do not participate in any covalent or ionic bond formation. Lone pairs therefore are called non-bonding electrons. But the lone pair being electrons and, therefore, negatively charged centres they participate in Hydrogen bonding as Hydrogen bond acceptors.
However, not all lone pair containing atoms become Hydrogen bond acceptors. Only those lone pair electrons closer to the nucleus and compact, as in N, O, and F, participate as H-bond acceptors. The diffused lone pair electrons of Cl, Br, and I do not show H-bonding except under exceptional circumstances. There are rare instances where Carbon, Chlorine, and Sulfur have shown Hydrogen Bonding.
Few other examples of Hydrogen bond donors and acceptors
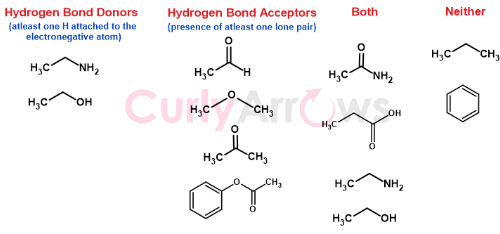
Subscribe to study Hydrogen bonding interactions, a stronger dipole-dipole Vander Waal force.
