There are three types of Van der Waal’s forces: Keesom Forces, Debye forces, and London Dispersion forces.
Keesom forces
Dutch physicist Willem Hendrik Keesom discovered that these forces occur in molecules with a polar covalent bond. The polarity difference is due to electronegativity, a property where one atom pulls more bond electrons leaving the other electron deficient. This creates two terminals- electron-rich (negative) and electron-deficient (positive). The negative end of the dipole interacts with the positive end of the neighboring polar molecule. All such interacting molecules orient themselves according to their poles leading to an orientation effect.
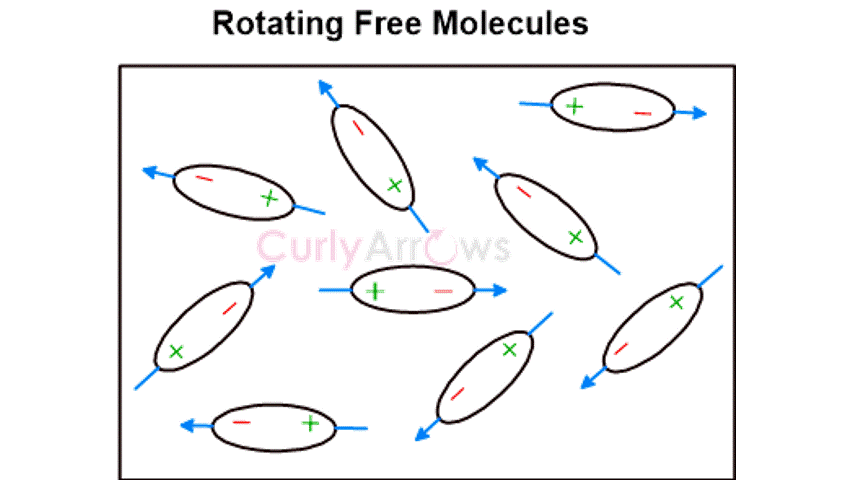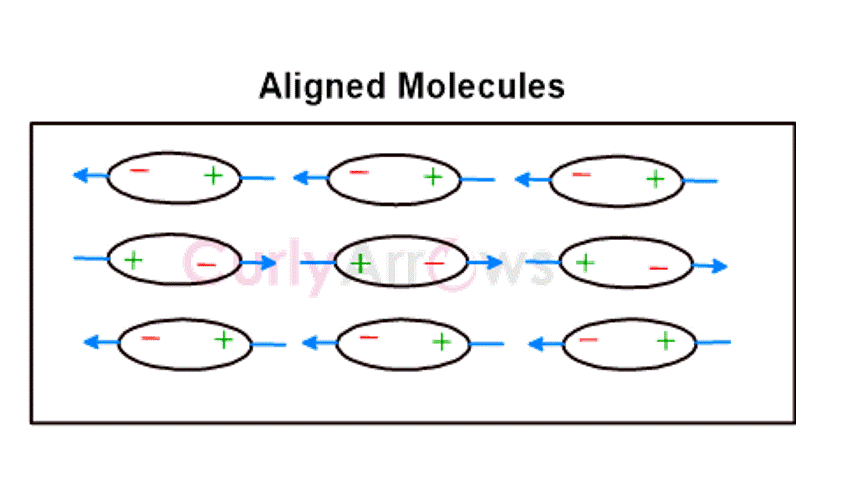
Examples of polar molecules are CO2, SO2, NH3, etc.
Debye Forces
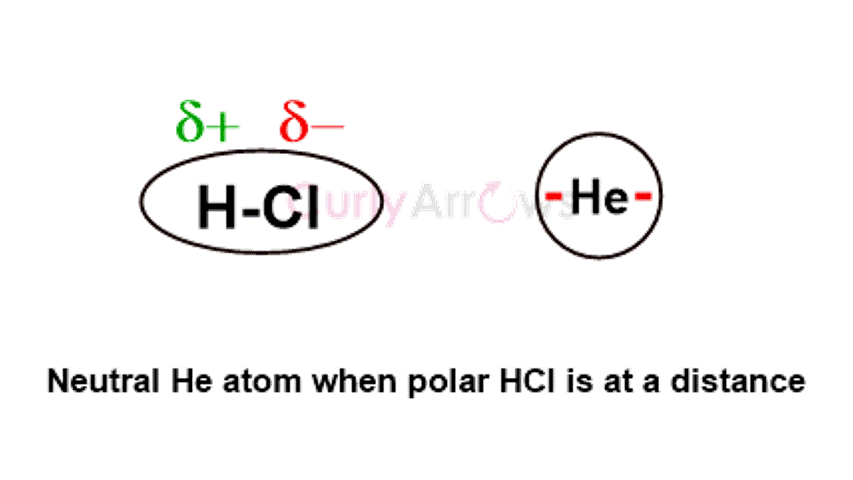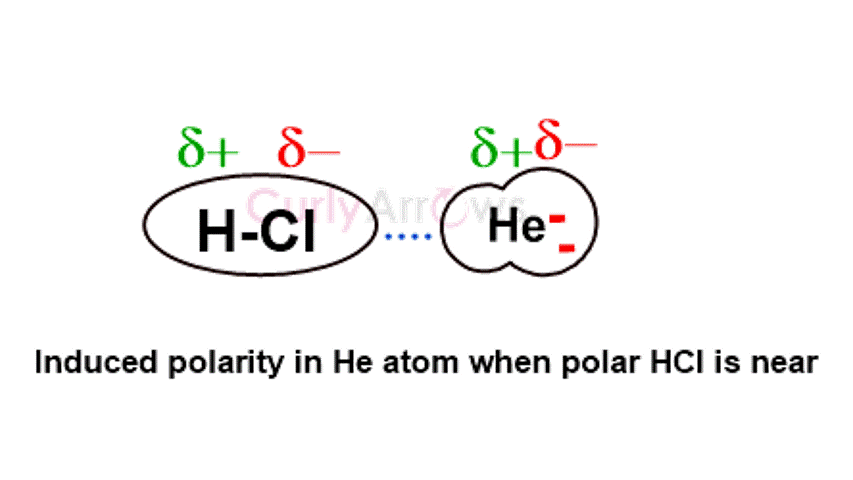
First identified by Dutch American physical chemist Peter Debye, these forces occur between polar and nonpolar molecules. When a nonpolar molecule comes in the vicinity of a polar molecule, due to its influence, the nonpolar molecule develops polar ends, leading to an induced polarity. Debye Forces are polar-nonpolar interactions arising from one’s permanent dipole and the other’s induced dipole.
London Forces
London Forces are the weakest intermolecular Van der Waal forces of attraction found in all atoms and molecules. It is named after the German physicist Fritz London and is also known as instantaneous dipole–induced dipole forces.
The continuous electron movement in an atom or a molecule creates a temporary dipole due to the disturbance in the electron arrangement.
The electron cloud becomes unsymmetrical, affecting nearby atoms and molecules and creating multiple induced dipoles. Such instantaneous dipole-induced dipole interactions are called London Forces.
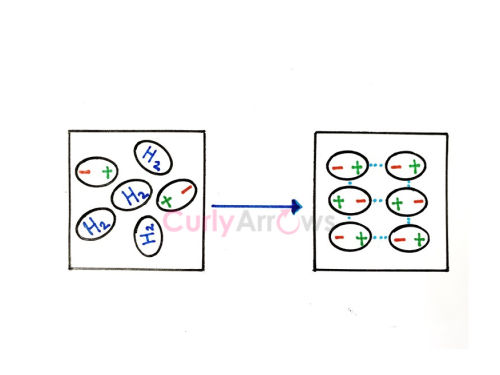
It causes nonpolar substances to condense into liquids and unite to a solid state when the temperature is lowered sufficiently.
For Example, H2 molecules associate with each other due to London forces where they form close dimers (H2)2 in Hydrogen gas.
This is a supportive content for the chapter Intermolecular Forces part of CurlyArrows' Introduction to Organic Chemistry Course. Preview the Book.
Related Reading-
Introduction to van Der Waal Forces
