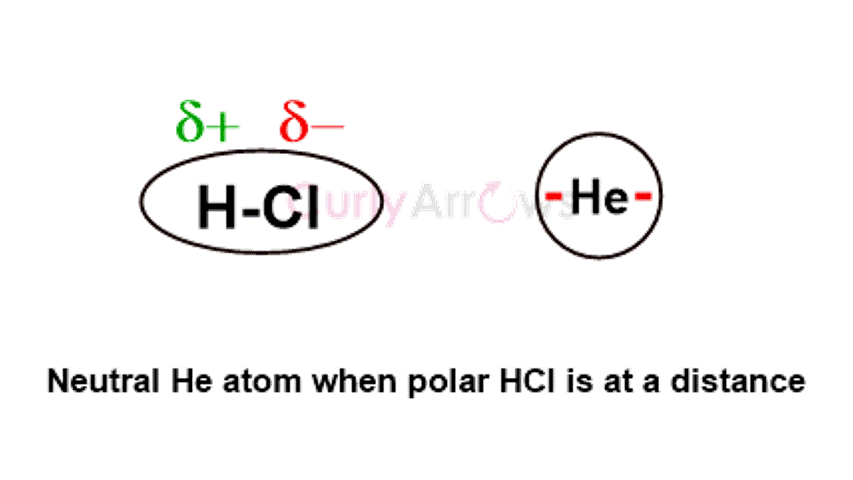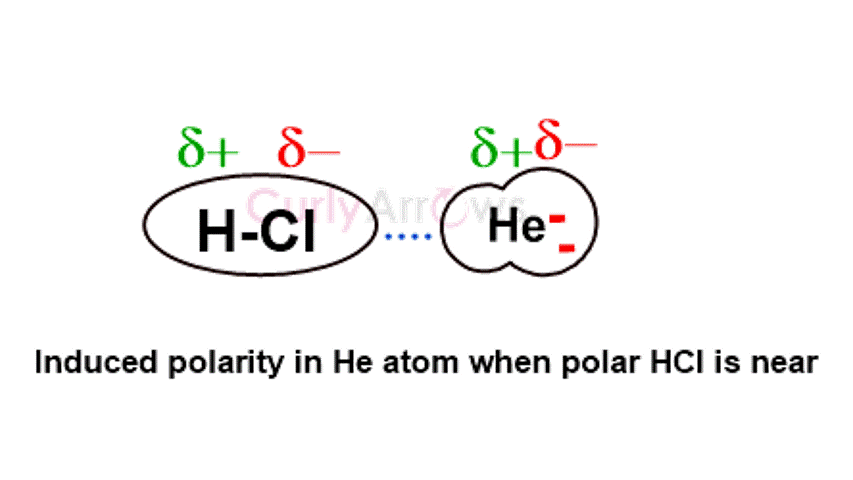
Debye intermolecular forces of attraction occur between polar and nonpolar atoms or molecules. When closely placed with a polar molecule (electronegativity difference between 0.5-1.7), the electron cloud of the nonpolar atom (or molecule) is influenced and distorted, leading to the formation of temporary dipoles. As the polarity is induced (forced) in the otherwise nonpolar atom, the Debye forces are said to show an Induction effect.
For example, HCl, a polar molecule, induces polarity in neutral, nonpolar He atom.
The above excerpt supports the chapter Intermolecular Forces part of CurlyArrows' Introductory Organic Chemistry Course. Preview the Book.
Related Reading- Vander Walls Polar-Nonpolar Interactions
