Learning Objective: To study Vander Waals Debye force, interactions, and the factors affecting their strength.
Skill Level - Intermediate
Prerequisites:
Chapter: Intermolecular Forces
Sub-topic: Van Der Waal Forces
Author's Note: There are two types of molecules – polar and nonpolar which can result in three types of intermolecular interactions: polar-polar, polar-nonpolar, and nonpolar-nonpolar. Based on these interactions, the Vander Waal forces are of three types, of which we cover the polar-nonpolar interactions in this section. Here, we will learn which molecules participate in such interactions and how to easily predict their strength by looking at two main factors – polarity and polarizability.
Vander Waals Debye (Polar-Nonpolar) Interactions
Debye, a subtype of Vander Waal's force, is an interaction between polar-nonpolar atoms or molecules.
An example is an interaction between a polar molecule like H-Cl and a neutral, nonpolar atom like Helium.
How Debye Interactions Occur?
Chlorine, the most electronegative atom in HCl, pulls and hoards the covalent bonds' electron density. The Cl end develops a partial negative charge (denoted as δ-) and becomes the negative terminal, whereas the Hydrogen end is positive (denoted as δ+). Therefore, the HCl molecule has two in-built polar ends (or a dipole) due to the Chlorine's electronegativity.
The dipole of HCl will induce pole formation in a neutral He atom when brought to its vicinity. The Helium atom's side closest to the Chlorine will develop an opposite positive charge; consequently, the other end will get a negative charge. Due to the induced polarity, they begin to attract.
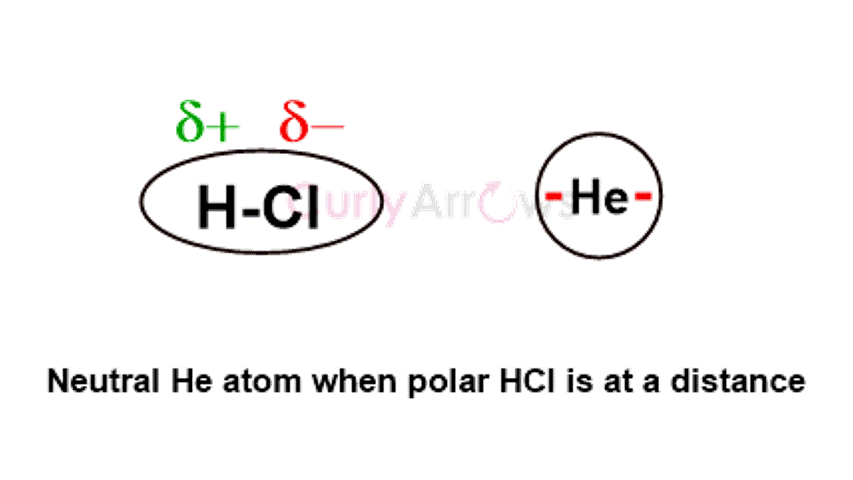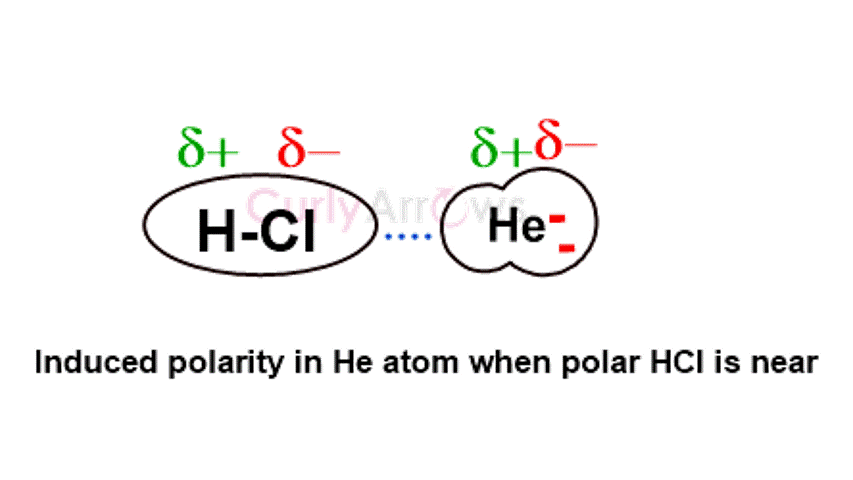
Polar HCl's impact on neutral Helium atoms can be better understood by considering an example of a magnet and a nail. The magnet's innate north and south poles can induce polarity in the ordinary iron nail.
In the magnet's presence, the iron nails achieve magnetism temporarily and begin to orient. The nails attract each other, similar to the attraction between HCl (as the magnet) and He atom (as iron nails).

Some other examples of polar-nonpolar molecular combinations are HF-H2, HCl-I2, and H2O-Ar.
Factors affecting the strength of Debye forces
The strength of polar-nonpolar molecular interactions will depend on two factors-
1) electronegativity of the polar molecule, and
2) polarizability of the neutral, nonpolar atom.
1) Electronegativity of the polar molecule
The greater the polarity of the polar molecule, the stronger will be the induced polarity and its subsequent interaction with the nonpolar molecule.
For example, comparing the strength of interaction between two polar molecules, HCl and HF, with the nonpolar He atom, it is observed that the Helium atom will interact more strongly with HF than with HCl since Fluorine is more electronegative than Chlorine.
Therefore, electronegativity is the power that will influence the electron cloud of the nonpolar atoms, distorting it and causing greater pole separation.
So, the HF-He pair will have the strongest attractive interaction than the He-HCl pair.

2) Polarizability of the neutral, nonpolar atom
The size of the nonpolar atoms considerably impacts the attractive intermolecular strength.
Bigger atoms have sizable electron clouds that can easily be influenced by nearby polar molecules, causing electron cloud distortion. The distortion allows the formation of poles; therefore, a nonpolar atom's size affects polarizability and increases the attractive strength.
For example, comparing the strength of interaction between HF, smaller Helium (He) atom, and larger Argon (Ar) atom, it is observed that the Argon atom interacts more strongly with HF than the Helium.

The Argon’s electrons being further away from the nucleus, are easily distorted to form temporary poles, resulting in stronger HF-Ar interaction.
Fewer Helium’s electrons are pulled by the nucleus strongly (nuclear charge), restricting its polarization.

Therefore, the bigger the atoms' size, the more the electron clouds, and the higher their molecular weight. Such atoms will have greater polarizability, translating to stronger intermolecular attractions.
Such polar-nonpolar interactions are also called the induction effect or dipole-induced dipole interactions.
Strength of Debye Interaction
The Debye force is stronger than the London force but weaker than the Keesom force.
Keesom force > Debye force > London force
Their other names-
Dipole-Dipole > Dipole-induced dipole > Induced Dipole-Induced Dipole
Orientation effect > Induction Effect > Dispersion effect

This excerpt is from the chapter Intermolecular Forces part of CurlyArrows' Introductory Organic Chemistry Course. Preview the Book. Subscribe.
Related concepts: Dipole-Dipole Vander Waal interactions, Induced dipole-induced dipole Vander Waal interactions.
