Accidental discoveries and insatiable curiosity fuel chemistry.
One such significant discovery is of isomerism, which also occurred around the period when the faith in vitalism, a belief that organic molecules must only come from living organisms, was dwindling.
It was a pleasant surprise to learn that urea, a by-product of urine produced by human kidneys, could also be synthesized by heating ammonium cyanate, an inorganic compound.

Even with this astonishing breakthrough in his pocket in 1828, Wöhler could not help noticing that the synthesized urea had the same chemical composition as ammonium cyanate, but they behaved quite differently.
Ammonium cyanate is an ionic salt, whereas urea is a covalent compound.
(Related Reading - Difference between organic and inorganic compounds)
Well, it was only a year before when he had prepared cyanic acid (HOCN) and had observed that although its elemental composition was identical to fulminic acid (HCNO), its properties were different.
With these discoveries came a shift in thinking about the needless involvement of a living being in producing organic molecules. The theory of vitalism was facing a downfall, and the field of organic chemistry was starting to take shape.
More observations of the same type were being reported elsewhere –
- In 1825, when Faraday was trying to prepare an illuminating gas by heating whale oil, he obtained an oily material consisting of two parts - a minor, liquid benzene (C6H6) and a major, volatile gas. The gas had the same percentage composition as ethylene (C2H4), though its density was double. Later, it was found that the compound was isobutylene (C4H8).
- Mitscherlich, in 1823, observed that sulfur crystals had different forms - monoclinic or rhombic.
- Aragonite and calcite were both crystalline forms of calcium carbonate. Iron pyrite and marcasite were both FeS2 and diamond and graphite were both carbon in composition.
- In 1822, Berzelius observed two different tin oxides with the same composition, and in 1825, he noted two forms of phosphoric acid with identical compositions.
- Around 1826, it was discovered that tartaric and racemic acids had identical compositions but some different properties.
Now, all these discoveries contradicted the law of definite proportion, which states that a substance's properties must correlate with its composition. This presented a real dilemma—how can two compounds with the same composition have different properties? Was the property a result of different arrangements of the elements?
It was J.J. Berzelius, Wohler’s Swedish teacher, who recognized such molecules in 1831-32 and named them isomers (Isomerisk in Swedish for equal parts). According to him, isomers have the same composition, being identical in their relative (for example, isobutylene and ethylene) and absolute (for example, cyanic acid and fulminic acid) numbers of atoms but having different properties. Berzelius believed that atoms within molecules can unite in different ways to produce isomers.
The concept of molar mass was still developing and raw.
After a while, the term isomer changed to include molar masses. It is now defined as compounds with equal composition and molar mass but different properties. Isomerism is the occurrence of such compounds.
The connectivity between atoms (constitution) became the first method to understand and classify isomers. Differences in connectivity between the atoms produced molecules with different structures and properties.
For example, as seen in the case of cyanic acid, H–OCN, and fulminate acid, H–CNO or between butane and 2-methylpropane.
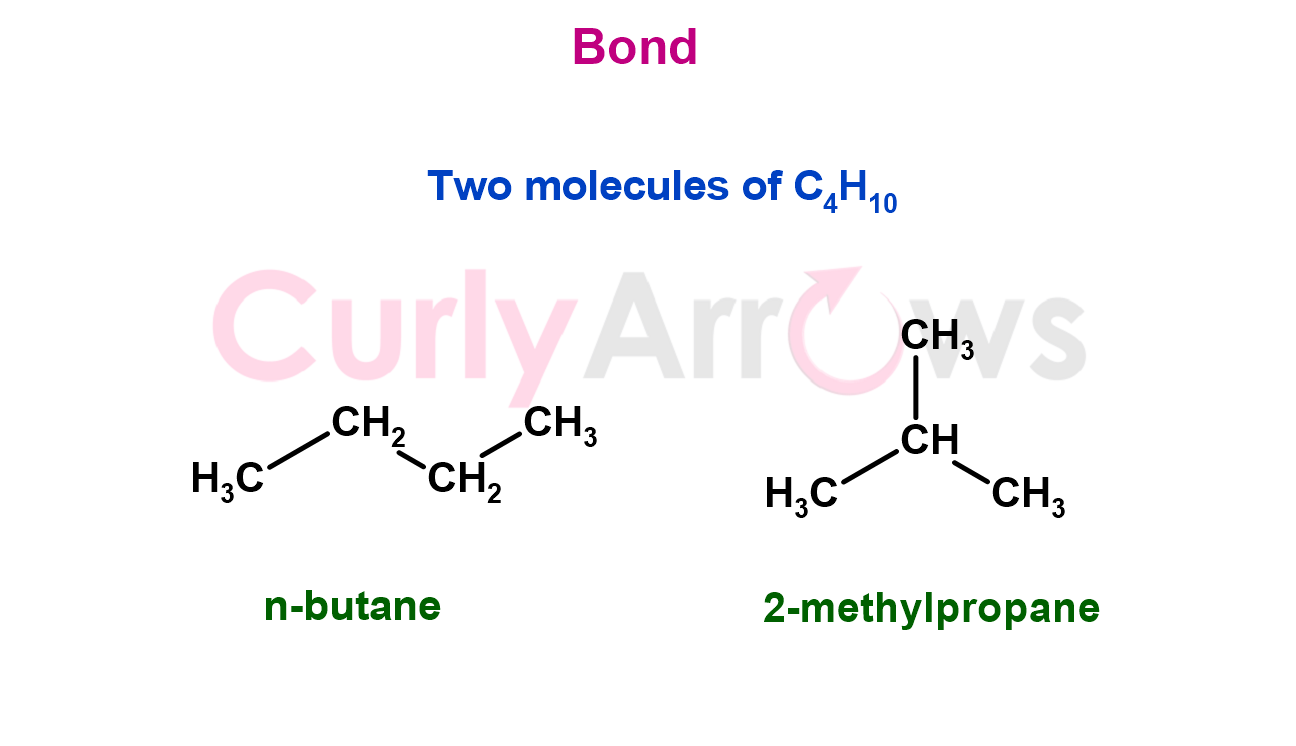
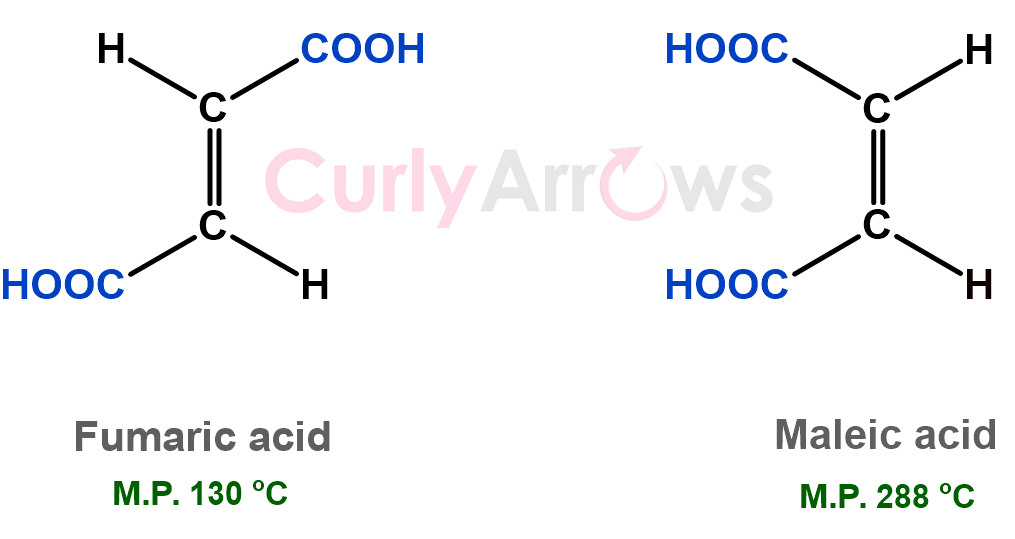
Later, molecules having the same connectivity were also found to show different properties. They differed in how those atoms were arranged in space relative to each other, beginning the era of stereochemistry.
Notice in the example below how the melting points changed with the change in the location of the -COOH group.
In some cases, molecules having the same connectivity and orientation in space were still different due to their temporary shape or conformation. The geometry of molecules changed due to their rotations along the single bonds producing conformation isomers having different geometries. The energy required for such changes in geometry between the molecules was low and occurred easily.
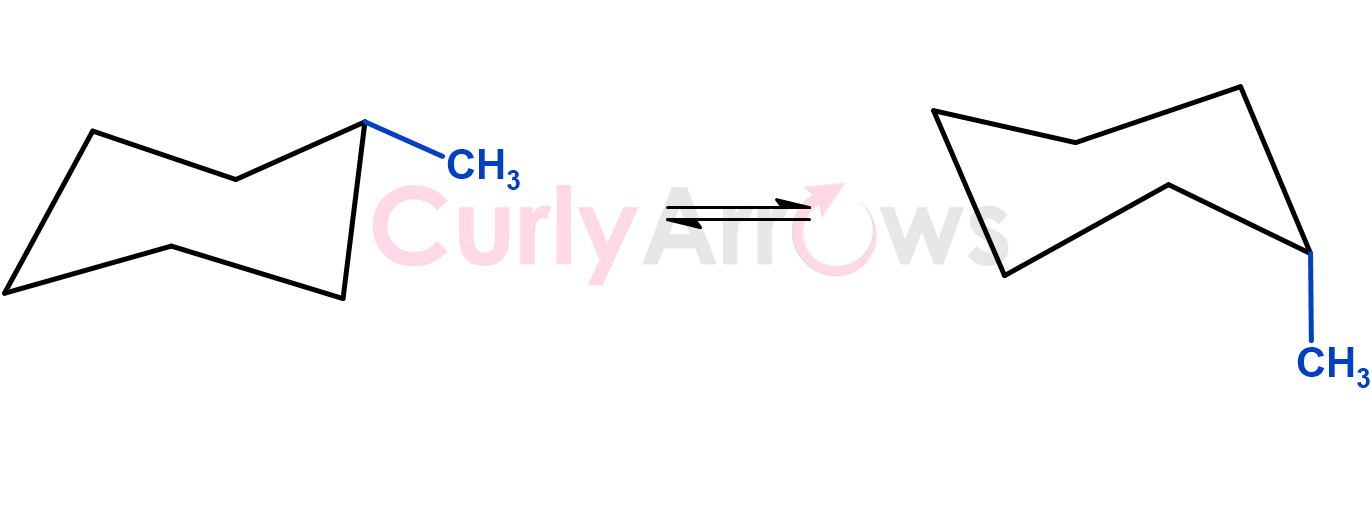
However, such changes were very important at the enzymatic level, where binding to an enzyme site to produce action required molecules to orient in the correct form.
Isomerism, as a developing and important concept, unveiled a multitude of isomers with different types, structures, and features. It became an important tenet in organic chemistry, opening a whole new chapter on structural isomerism and establishing structural formulas. It now became easier to classify organic compounds based on their structures, making organic chemistry clearer and, in the words of Wöhler, not a “dark forest with few or no pathways.”
References:
Liebig–Wöhler Controversy and the Concept of Isomerism
Jacob Berzelius Contribution to Chemistry
