Learning Objective: To study what Van Der Waal forces are in chemistry and their example.
Skill Level - Intermediate
Prerequisites:
Chapter: Intermolecular Forces
Sub-topic: Van Der Waal Forces
Author's Note: Not all interactions between molecules are bond-forming. Several times they interact to form cohesive units, where their collective strength is responsible for bringing shape, properties, and strength. Think water droplets on a leaf, the helical shape of DNA, compressed liquefied gases, and the strength of polymer sheets. All these ubiquitous interactions are attributed to the Van Der Waal Forces, named after the Dutch physicist, Johannes Diderik van der Waals. This section introduces Van Der Waal Forces and its subtypes and illustrates them with many examples.
Introduction to the Vander Waals Forces (Chemistry)- With Examples
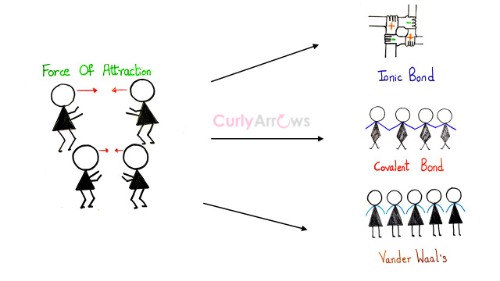
Attractive forces bring the atoms, molecules, and ions close. If their interactions are strong, they can form bonds like ionic and covalent. However, if the attractive force is weak, in that case, the molecules or atoms group closely without forming any actual bonds or linkages. Such a weak intermolecular attraction is said to occur by the van der Waals force.
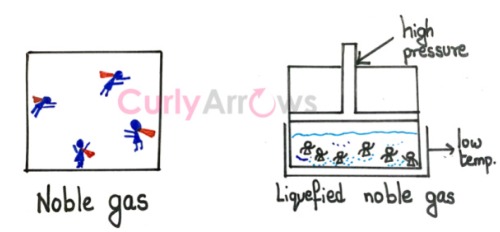
Since the van der Waals forces are attractive, they are distance-dependent. It gets weaker and non-existent as the distance between the atoms or molecules increases.
Also, the participants must be neutral atoms, polar and nonpolar molecules, but not ions. Since ionic interactions also depend on the force of attraction, they are stronger than the van der Waals forces and are studied separately.
Dutch physicist Johannes Diederik van der Waals discovered these attractive intermolecular forces while researching the ideal gas law.
Future investigations revealed three types of Vander Waals interactions - Debye, Keesom, and the London force.
The London Dispersion force is the weakest van der Waal force but is the most universal.
Examples of van der Waals’ Force
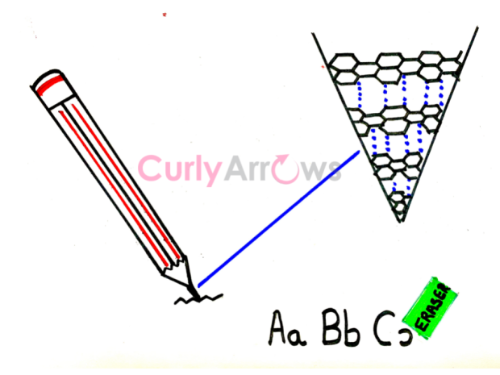
1) Graphite, a form of Carbon seen in a pencil, is a good example where one observes van der Waals' force. The carbon layers in graphite are neatly stacked one on top of the other due to Vander Waals' attraction and slide effortlessly onto the paper while writing. Again, these layers can be easily picked up by an eraser due to van der Waal's attraction to leave behind a blank surface.
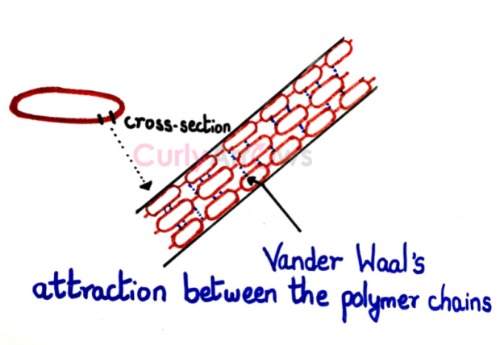
2) A rubber band has long chains of repeating molecular units that are attracted to each other using the Vander Waals force, allowing the rubber band to stretch and recoil.
3) In humans, van der Waals’ force manifests in blood circulation. Blood, comprising many cell components, flows effortlessly across our bodies without sticking or holding onto the surfaces of tissues and organs.
4) The lizards crawl on the wall using Vander Waal’s adhesive strength, defying gravity.

This is an excerpt from CurlyArrows' Introduction to Organic Chemistry Premium Tutorials, Chapter Intermolecular Forces. Subscribe.
