Van der Waals forces are weak intermolecular attractive forces that occur in polar and nonpolar atoms or molecules due to the shift in their electron positions.
The electrons shift to form electron-dense and electron-deficient poles. Some poles are permanent due to the nature of the atom in a molecule, while others are induced poles.
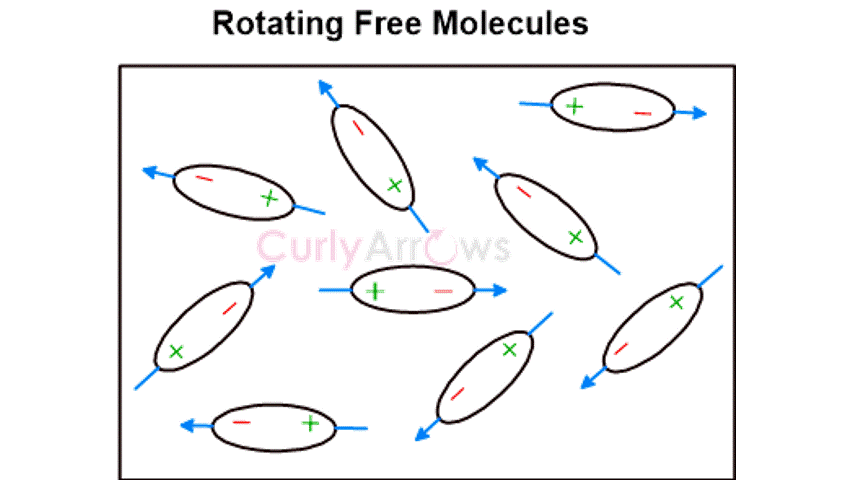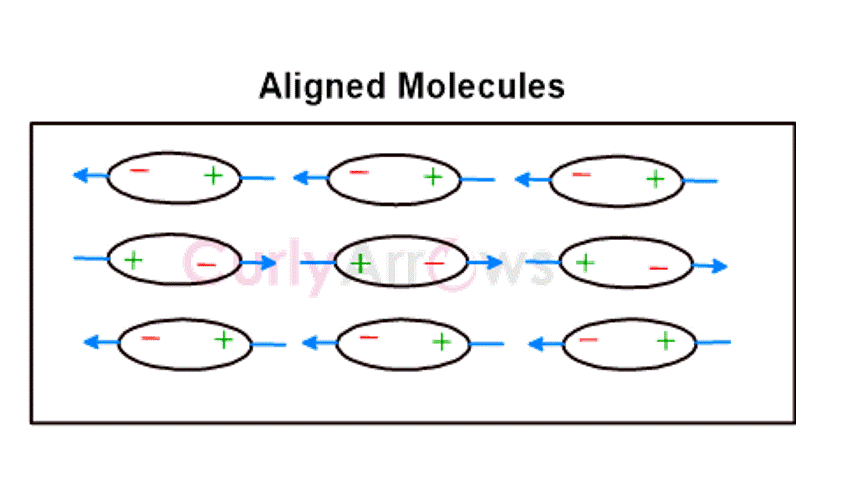
The opposite poles of different molecules then attract each other to form cohesive molecular units. Their attraction is strong when the distance between the atoms and the molecules is smaller. This kind of attractive van Der Waal molecular interaction is the most basic and universal way by which all atoms and molecules interact.
Breaking these cohesive molecules requires energy, and depending on the strength of the interaction, the energy requirement increases. Therefore, van Der Waal's forces affect molecules' physical properties of boiling point, melting point, and viscosity.
The above excerpt supports the chapter Intermolecular Forces part of CurlyArrows' Introductory Organic Chemistry Course. Preview the Book.
Related Reading - Introduction to the van Der Waal Forces