Learning Objective: To learn about ring–chain isomerism, a subtype of structural isomerism.
Skill Level – Intermediate
Prerequisites:
Double Bond Equivalent – How to find and interpret DBE values?
IUPAC writing guidelines
Related –
Chapter: Structural Isomerism
Author's Note: Ring-chain isomerism demonstrates two forms of representing an organic compound that can exist as a flexible open chain or rigid close structure. This difference leads to various possibilities –
- Certain drugs or natural compounds may exhibit different effects depending on whether they are in an open-chain or ring form.
- Open-chain compounds may be more reactive in specific reactions than their ring counterparts, which can be more stable.
- The physical properties of ring-chain isomers, such as boiling points, melting points, and solubility, can differ significantly, affecting separation techniques.
Therefore, it is essential to study how to identify two ring-chain isomers and predict their structural formula. This section details ring-chain isomerism, its examples, functional groups commonly exhibiting ring-chain isomerism, and more.
Ring - Chain Isomerism
Compounds may have the same molecular formula but can vary in their structures such that they can exist in open chain or closed are called ring-chain isomers, and the phenomenon is called ring-chain isomerism.
The compounds must have at least a DBE value of 1 to show ring and chain isomerism.
For example,
a) Alkenes and cycloalkanes (CnH2n)
An example of CnH2n is C3H6, a molecular formula that has one degree of unsaturation in this structure.
One degree of unsaturation (*DBE) can be one double bond or a cyclic ring. Therefore, the ring-chain isomer of C3H6 is-

C4H8 represents-
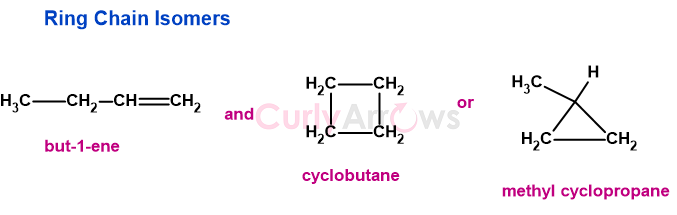
b) Alkynes and cycloalkenes (CnH2n-2)
An example of CnH2n-2 is ....
c) Unsaturated alcohols and cyclic ethers (CnH2nO)
Molecules with the general molecular formula CnH2nO have....
Subscribe to learn all subtypes of structural isomerism.
