Keesom forces occur in polar molecules where the atoms have a considerable electronegativity difference, resulting in the charge separation and formation of positive and negative poles. The negative pole of one molecule electrostatically interacts with the positive pole of the neighboring molecule. Consequently, all the polar molecules in the medium try to orient their dipoles to be in alignment for such electrostatic interactions. This effect is called the Orientation effect . Examples are HCl and NH3,etc.
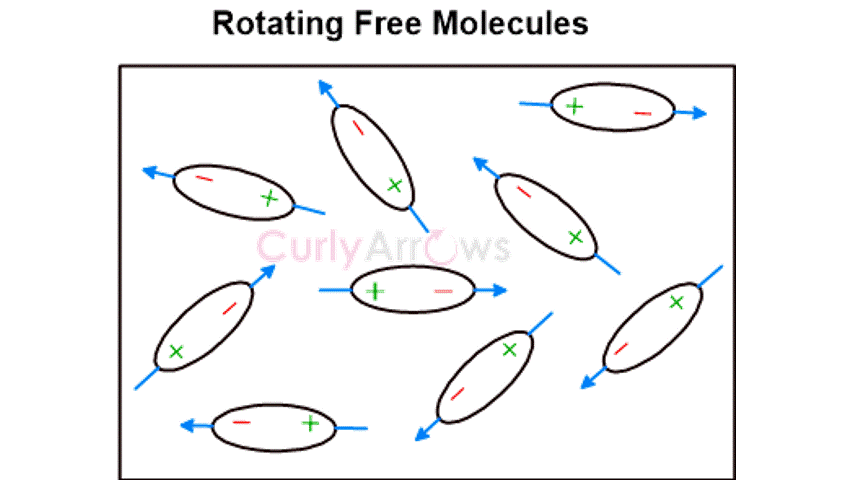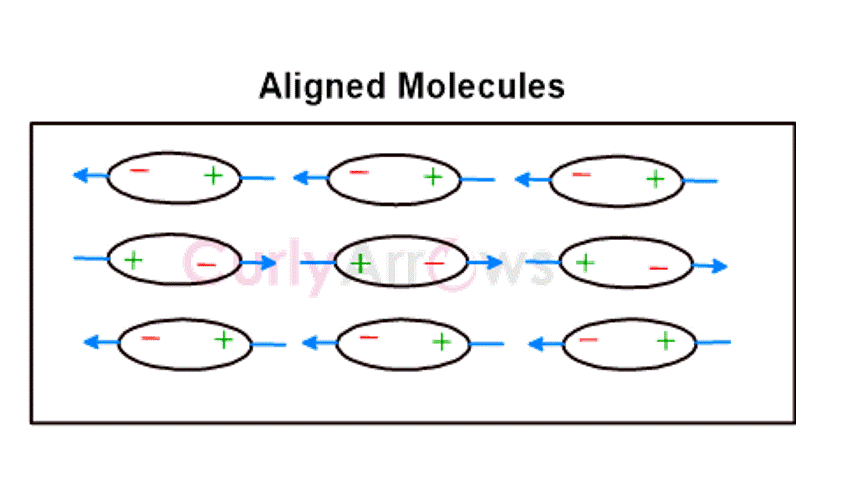
This is a supportive content for the chapter Intermolecular Forces part of CurlyArrows' Introduction to Organic Chemistry Course. Preview the Book.
Related Reading-
