Learning Objective: To study Hydrogen bonding interactions, a type of dipole-dipole Vander Waal forces.
Skill Level – Intermediate
Prerequisites:
Difference between Ionic, covalent, Metallic, and Vander Waal Forces
Related Concept – Physical Properties affected by the lone pair | Hydrogen Bonding, Shape and Bond Angle, Polarity, and Dipole Moment
Chapter: Intermolecular Forces
Sub-topic: Hydrogen Bonding
Author's Note: Students often get confused about whether or not Hydrogen bonding is a Vander Waal interaction. Hydrogen bonding is, in reality, a stronger form of dipole-dipole Vander Waal forces. However, Hydrogen bonding interactions are treated separately from other Vander Waal forces since it is only seen in some molecules that possess electronegative atoms N, O, or F.
Still, the interacting molecule must meet certain requirements of having a Hydrogen bond acceptor atom, Hydrogen bond donor atoms, and be at a collinear bond angle for maximizing the benefits.
This section mainly introduces Hydrogen bond interaction, how to visualize a hydrogen bond, how it occurs, and a few examples of molecules that show Hydrogen bonding.
The utility of Hydrogen bonding interactions extends beyond the simple solubility applications in chemistry to biology, physics, and several other aspects of human life. Next time you see whipped cream frosting on your cupcakes, remember that such air bubbles and heavy cream could only stabilize and create that fluffy structure due to Hydrogen bonding interactions. Otherwise, the whipped cream would weep.
Hydrogen Bonding
The bond-forming interactions between atoms of a specific kind can form various ionic, polar, and nonpolar covalent molecules.
Of these, the polar and nonpolar covalent molecules can further electrostatically interact with each other without forming any new bonds. These non-bonding interactions are called Vander Waals that control many physical, chemical, and biological properties of shape, nature, reactivity, etc.
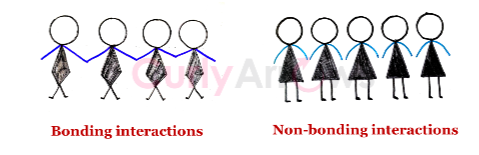
Discoveries didn’t end at that point. Scientists in 1912 found a different type of new non-bonding molecular interaction potent than the Vander Waals interaction exhibited only by the polar covalent molecules. The interaction was specifically active when the polarity in a molecule is due to electronegative atoms (N, O, or F) to which Hydrogen is covalently bonded. This interaction is called Hydrogen bonding and affects various other properties of nature, shape, solubility, density, melting point, and boiling point of a compound.
For example, water is liquid in nature at room temperature due to the hydrogen bonds of the water molecules (H2O), which water’s closest cousin, H2S, does not show. This close association is hard to break; therefore, water boils at a high temperature of 100oC to free the linked H2O into individual molecules.

DNA has two shapes - helical and pleated due to the Hydrogen bonds holding nucleic acid strands, thereby influencing their forms.

Organic and few inorganic acids and alcohols show high solubility in water due to their ability to form Hydrogen bonds.

HF - an inorganic compound exists in a liquid state due to Hydrogen bonding, whereas its counterparts HCl, HBr, HI are gaseous.

The crystalline ice has water molecules held in a hexagonal shape due to the Hydrogen bonds that create spaces affecting the density, and that floats ice on the water.

What is Hydrogen Bonding ?
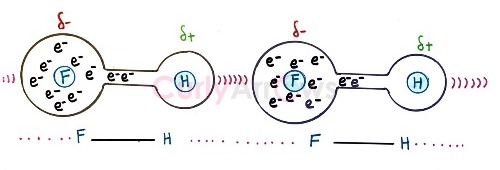
Hydrogen bonding is a type of intermolecular interaction that specifically occurs when the Hydrogen is between highly electronegative atoms N, O or F. The Hydrogen is covalently bonded to one electronegative atom, and it tries to pull by attraction the other neighbouring electronegative atom without forming any formal bond. This newly formed attractive interaction is called the Hydrogen bond and is shown as a dash (---).
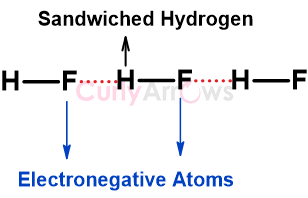
Visually, the electronegative atoms will look like the terminals of the linear bridge with Hydrogen atom holding the two ends.
However, unlike in the bridge the Hydrogen may or may not be equidistant from the two electronegative atoms. The equidistant property would depend on the nature of the electronegative atoms involved.
Why Hydrogen Bonding occurs?
Hydrogen is unique to have only one electron in the 1s orbital, the electron position closest to the nucleus. When Hydrogen shares this 1s electron to form a covalent bond with electronegative atoms N, O and F, it exposes the nucleus of the Hydrogen, a highly electron-deficient region.
For example, the covalent bond of the HF molecule is formed using one electron of Hydrogen.
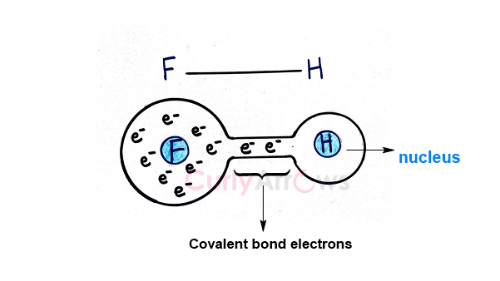
Not only has the Hydrogen lost its only electron to the covalent bond formation, but the electronegative atom Fluorine (F) also pulls away most of the bond electrons towards itself.
By stealing most of the Hydrogen atoms electron density, the Fluorine (F) gets a partial negative charge (δ-), and the Hydrogen gets a partial positive charge (δ+).
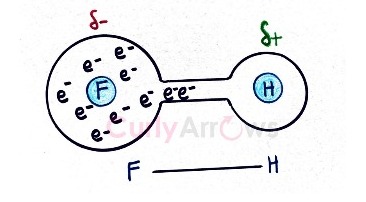
The Hydrogen atom is now highly electron-deficient, and it tries to pull the electrons from the neighbouring electronegative atom by attraction. This creates an inter molecular interaction called the Hydrogen bond and is denoted by a dotted line (…).
Hydrogen bonding is a special case of dipole-dipole interaction; however, it is only seen in electronegative atoms containing at least one active lone pair. Lone pairs are unused electrons from bond formation reactions; however, being electrons and negatively charged centres, they participate in Hydrogen bonding as Hydrogen bond acceptors.
(Related Concept - Lone pair)
If one tries to visualize the hydrogen bonding intermolecular interaction mentally, it will look a lot like a hook-and-loop process.
The loss of electrons to the bonded electronegative atom prompts the electron-deficient Hydrogen to throw a hook at the neighboring electronegative atom’s loop (electron-rich lone pair on N, O, or F atoms) to pull it closer and interact.
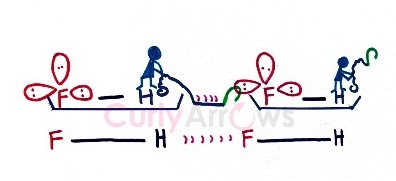
Such an electrostatic interaction is more potent than Vander Waals but weaker than covalent and ionic bonds.
Next: The donors and acceptor atoms in the Hydrogen Bond
Next: Hydrogen Bond Strength, Significance, and Types
Next: Factors affecting Hydrogen Bond Strength
Next: Impact of Hydrogen bonding on Physical Properties - Melting and boiling point, Solubility, and State
Next: Calculate the number of active hydrogen bonds per molecule
Take the Quiz
