From the Chapter - Electronic Displacements in a Covalent Bond
Carbon and Hydrogen do not have any significant electronegativity difference (< 0.5) and can form long chains of neutral hydrocarbon compounds like Butane, Pentane, Hexane, Heptane, Octane, etc.
But suppose at one end of a carbon-carbon chain, there lies an atom or a group of atoms with the ability to pull the bond electrons towards itself or push the bond electrons from itself, and decreasingly transmit the effect to the adjacent carbons via the covalent bonds. In that case, such an effect is called an Inductive effect.
How the Inductive Effect Occurs
Electronegativity is an important factor that drives electron shifts. Higher electronegative heteroatoms (N, O, S, Halogens, etc.) in the hydrocarbon chain introduce polarity difference between the Carbon and the heteroatom. This polarization further induces polarization in the adjacent carbon atoms, and its effect weakens with the distance. The effect is negligible after the fourth carbon atom in a longer chain.
For example, the structure of n-butane (made of four Carbon atoms) is C4H3-C3H2-C2H2-C1H3, and if one of the terminal Hydrogen of C1 is replaced with an electron-withdrawing group, nitro (-NO2), the structure is C4H3-C3H2-C2H2-C1H2-NO2.
The replacement of H with more electronegative group nitro (-NO2) pulls the C-NO2 bond electrons towards NO2, creating a polarization of the C-NO2 bond. Since more significant electron density resides on NO2, it gets a partial negative charge (δ-), and Carbon, due to the loss in electron density, gets a partial positive charge (δ+).
C4H3-C3H2-C2H2- δ+C1H2- δ-NO2
The loss in the electron density of C1 causes it to pull the electron density from C2, so C2 is now electron deficient. This displacement of electrons of the C2-C1 bond towards C1 and polarization of C2 is shown with one more δ+ charge as δ+ δ+C2.
C4H3-C3H2- δ+ δ+C2H2- δ+C1H2- δ-NO2
Pulling the bond electrons from C2 makes it slightly electronegative and partially polarized, causing C2 to withdraw the electron density even slightly from C3. The C3 will, in turn, pull from C4, and the bond polarization is shown as,
δ+ δ+ δ+ δ+C4H3- δ+ δ+ δ+C3H2- δ+ δ+C2H2- δ+C1H2- δ-NO2
The effect of the induced polarity steadily weakens with the increasing distance from the substituent and becomes negligible after three-four Carbon atoms. In the above example, the impact is felt slightly or is insignificant for C4.
In short, the adjacent carbon atoms attained induced electronegativity due to the electronegative atom, and polarizations got relayed through the single covalent bonds until the effect diminished with the distance.
The Inductive effect depends on the electron- withdrawing/donating nature of the atoms present and is therefore inherent to the molecule. It is a permanent effect and does not require the presence of any reagent or chemical environment.
Types of Inductive Effect
The Inductive effect is of two types:
1) -I effect (Electron Pulling)
If the atom or the group of atoms pulls the bond electrons, then such an effect is called a negative Inductive effect or the -I effect. For example, in CH3-CH2-CH2-Cl, electronegative Chlorine withdraws the electron density from the carbon-carbon bonds.
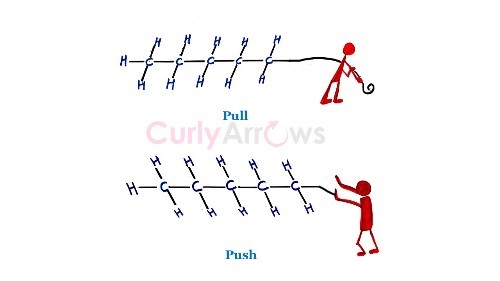
The ‘pull’ effect of an electron-withdrawing group (-I effect) is similar to the game of tug-of-war. The electron-withdrawing group tug on the bond electrons of the rope. The tugging is felt strongly by the nearby atoms and less by the faraway atoms until the central bond electrons move towards the electron- withdrawing group.
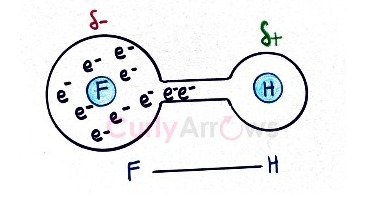
For example, in the above image, the Fluoro functional group pulls the two bond electrons of the H-F bond towards itself and therefore displays -I effect.
2) +I effect (Electron Pushing)
If the atom of the group of atoms pushes the bond electrons, then the effect is called a positive Inductive effect or the +I effect .
For example, in toluene (CH3-C6H5), CH3 is less electronegative than Ar (according to the decreasing order of electronegativity, table below). Therefore, the methyl group (-CH3) pushes the electron density to the Phenyl (-C6H5) ring where Phenyl is acting as a -I group.
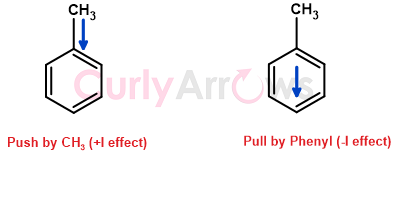
The push effect of an electron-donating group (+I effect) is like the release of energy seen in earthquakes where the surroundings near the epicentre has the most impact than the peripheral regions. The electron-donating groups push the electrons along the carbon chain, and its effect is weakest for the far-away carbon atoms.
Representation of Inductive (+I/-I) Effect
The arrow on top of the bond indicates the direction of pull or the push of the bond electrons.
The arrowhead pointing towards the electron-withdrawing group indicates the pull towards it.
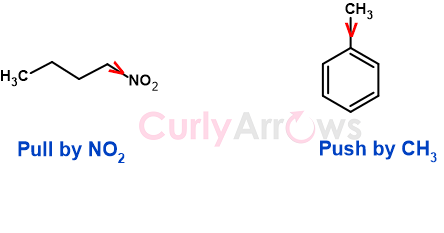
The arrowhead pointing away from the electron-donating group indicates the push of the bond electrons.
Functional Group Classification by Inductive Effect
All the groups of atoms are classified as electron-withdrawing (-I) or electron-donating (+I) relative to Hydrogen.
The most common +I and -I functional groups are:
+I group | -I group |
|---|---|
O- | OH, OR, OAr, Ar |
COO- | COOH, COOR |
CH3, CHR2, CH2R, CR3 | NO2, CN |
D | NH2, NH3+, NR3+ |
| F, Cl, Br, I, CF3 |
| SH, SR, SR2+, SO2R, SO2Ar |
| C_=CR, CH=CR2 |
| CHO, COR |
The -I effect of some of the atoms and groups in the decreasing order to Hydrogen is:
NH3+> -NO2> -CN> -SO3H> -CHO> -CO-> -COOH> -COCl> -CONH2> -F> -Cl> -Br> -I> -OH> -OR> -NH2> -C6H5> -CH3> -H
The +I effect of some of the alkyl group of atoms in the decreasing order to Hydrogen is:
-COO-> R3C- > R2CH-, RCH2-> CH3-> D-> H-
The position of a group in either order reflects how strongly it can withdraw or donate electron density through the bonds of a carbon chain relative to hydrogen.
Role of Inductive Effect in Organic Chemistry
Inductive effect, a foundational concept in organic chemistry operates through single covalent bonds alone, making it a permanent and inherent property of every organic molecule that contains a heteroatom or a polar substituent. Whether a substituent displays a –I effect or a +I effect depends entirely on its electronegativity relative to carbon. Electron-withdrawing groups — halogens, nitro groups, carbonyl-containing substituents — create regions of electron deficiency along the carbon chain, while electron-donating alkyl groups push electron density outward. Because this polarization decreases with each successive carbon atom and becomes negligible beyond three to four bonds, the inductive effect is most significant when the substituent is close to the reactive site of the molecule.
Understanding the inductive effect is essential for predicting a wide range of chemical behaviour in organic chemistry: the relative acidity of carboxylic acids, the stability of carbocations and carbanions, the basicity of amines, and the reactivity of substrates in substitution and elimination reactions. Coupled with bond polarity, electronegativity, and the –I/+I classification of functional groups chemists get a reliable framework for reasoning about electron distribution — and therefore reactivity — across an enormous range of covalent compounds.
This is an excerpt from CurlyArrows' Introduction to Organic Chemistry Premium Tutorials, Chapter Electronic Displacements in a Covalent Bond.
Frequently Asked Questions
The –I effect (negative inductive effect) occurs when an atom or group pulls bond electrons towards itself — an electron-withdrawing effect. Halogens such as fluorine and chlorine, nitro groups, and carbonyl-containing groups are classic –I groups.
The +I effect (positive inductive effect) occurs when an atom or group pushes bond electrons away from itself — an electron-donating effect. Alkyl groups such as –CH₃ are typical +I groups. All functional groups are classified as either –I or +I relative to hydrogen.
No — they are related but not the same. Electronegativity is the inherent ability of an atom to attract electrons in a bond. The inductive effect is the consequence of that electronegativity difference: the transmission of polarity through successive sigma bonds along a carbon chain.
Electronegativity is a property of an individual atom; the inductive effect is a molecular property arising from electronegativity differences between atoms. Higher electronegative heteroatoms such as N, O, S, and halogens introduce a polarity that propagates through the chain — that propagation is the inductive effect.
An arrow is drawn on top of the bond to indicate the direction of electron movement:
- When the arrowhead points towards an electron-withdrawing group → –I effect (electrons being pulled in that direction).
- When the arrowhead points away from an electron-donating group → +I effect (electrons being pushed outward).
Functional groups ranked in decreasing order of –I effect relative to hydrogen:
NH₃⁺ > –NO₂ > –CN > –SO₃H > –CHO > –CO– > –COOH > –COCl > –CONH₂ > –F > –Cl > –Br > –I > –OH > –OR > –NH₂ > –C₆H₅ > –CH₃ > –H
Groups higher in this order withdraw electron density more strongly through the sigma bonds of a carbon chain.
Electron-donating groups that show the +I effect include O⁻, COO⁻, and alkyl groups such as –CH₃, –CH₂R, –CHR₂, and –CR₃. The decreasing order of +I effect relative to hydrogen is:
–COO⁻ > R₃C– > R₂CH–, RCH₂– > –CH₃ > D > H
The position of a group in either order reflects how strongly it can withdraw or donate electron density through the bonds of a carbon chain relative to hydrogen.
–I effect → tug-of-war. The electron-withdrawing group tugs on the bond electrons like a rope. The tugging is felt strongly by nearby atoms and progressively less by faraway atoms, until the electrons shift towards the withdrawing group.
+I effect → earthquake. The electron-donating push is strongest near the epicentre (the substituent) and weakens the further along the chain you move — just as seismic impact diminishes with distance from the source.
The inductive effect is essential for predicting a wide range of chemical behaviour, including:
- The relative acidity of carboxylic acids
- The stability of carbocations and carbanions
- The basicity of amines
- The reactivity of substrates in substitution and elimination reactions
Coupled with bond polarity and electronegativity, the –I/+I classification of functional groups gives chemists a reliable framework for reasoning about electron distribution — and therefore reactivity — across an enormous range of covalent compounds.
Related Reading: Is the Inductive effect the same as Electronegativity? (Free)
