Pre-requisite Article- What is a Hydrogen bond?
The Hydrogen covalently bonded to an electronegative atom is called the donor atom. The donor atom is always found in between the two electronegative atoms.
The acceptor atom is the other electronegative atom that electron starved Hydrogen tries to attract.
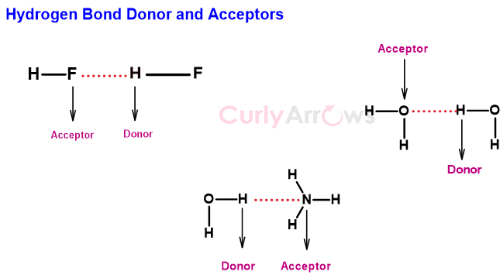
Though the name is Hydrogen bond donor, the molecule does not donate the Hydrogen as protons but barters its positive charge for the electron density of the neighboring electronegative atom. At the same time, it maintains its covalent bond character.
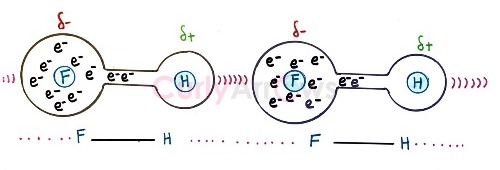
Therefore, a Hydrogen bond donor must have at least one Hydrogen atom in their molecule. Few examples of molecules that are Hydrogen bond donors are- NH3, H2O, HF, ROH, NH(R2), NH2R.
The Hydrogen bond acceptor is also an electronegative atom -N, O, or F. The acceptor atom must at least possess one lone pair electron to contribute to the electron-deficient Hydrogen of the donor atom.
However, not all lone pair containing atoms become Hydrogen bond acceptors. Only those lone pair electrons closer to the nucleus and compact, as in N, O, and F, participate as H-bond acceptors. The diffused lone pair electrons of Cl, Br, and I do not show H-bonding except under exceptional circumstances. There are rare instances where Carbon, Chlorine, and Sulfur have shown Hydrogen Bonding.
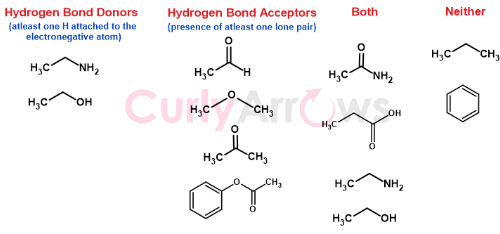
Few other examples of Hydrogen bond donors and acceptors