Curly Arrows - the curved and barbed arrows you might have seen in reactions have been the language of organic chemistry for over 100 years.
Fundamentally, the arrows represent the path of the electrons. The way they hop between atoms dictates whether a bond will form or break, causing those miraculous molecular transformations.
There are five main rules to consider while drawing reaction mechanisms using double-headed curly (or curved) arrows in organic chemistry. They are -
1) First, the tail of the arrow always points to an electron-rich centre, which could be either a bond or a lone pair.
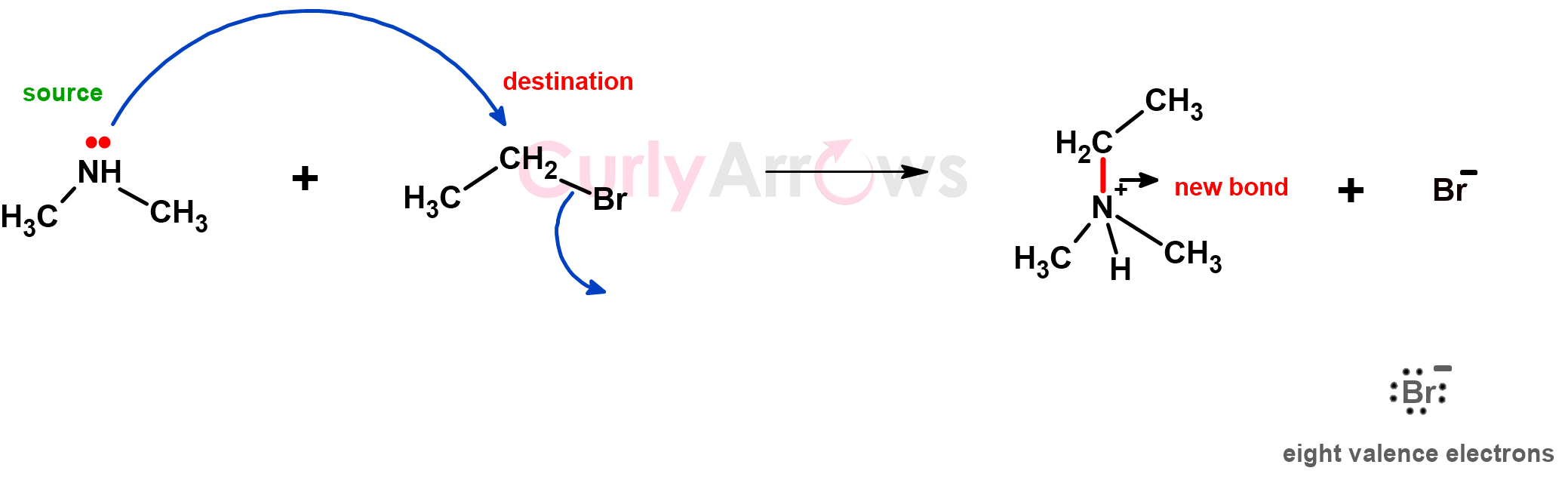
After heterolytic bond cleavage, an excess of electrons on an atom is shown as a negative charge, as in Br-.
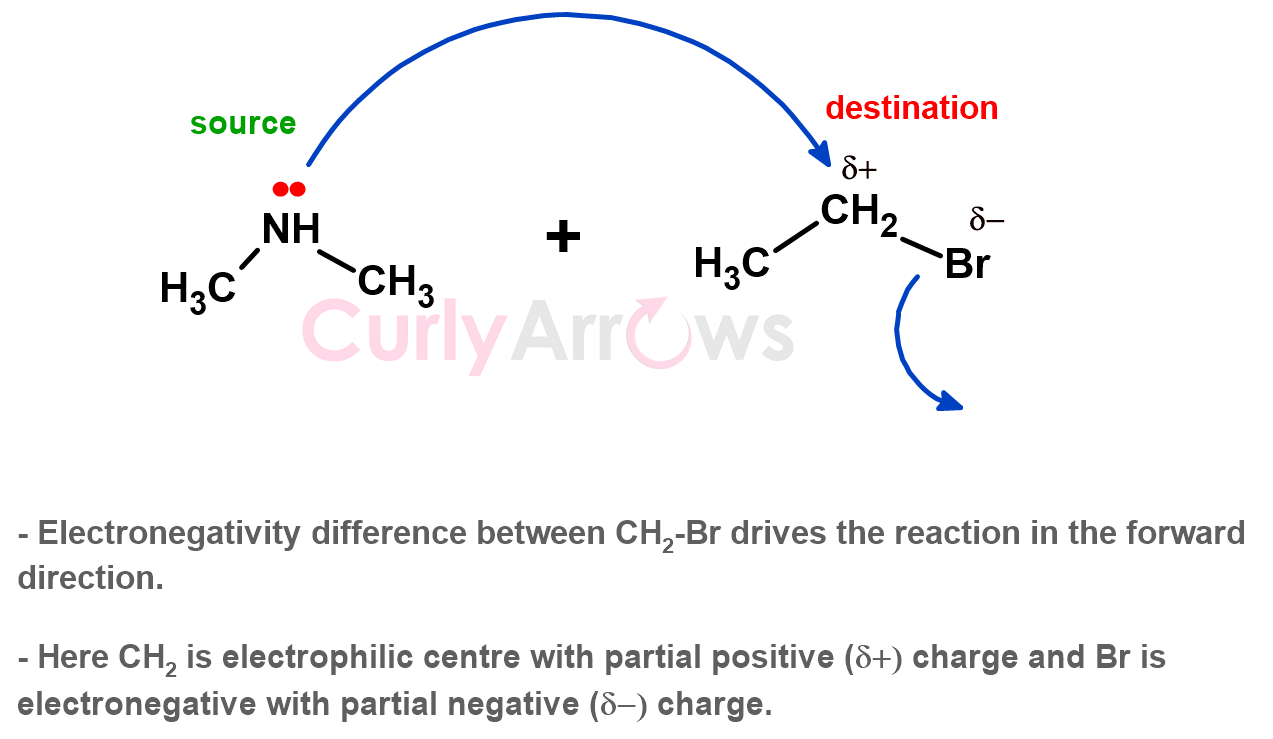
2) Second, the arrowhead points to the electron-deficient centre that receives those electrons. It is easily identifiable by its full or partial positive sign. Positive signifies a deficit of negative electrons or an imbalance of bond electron density.
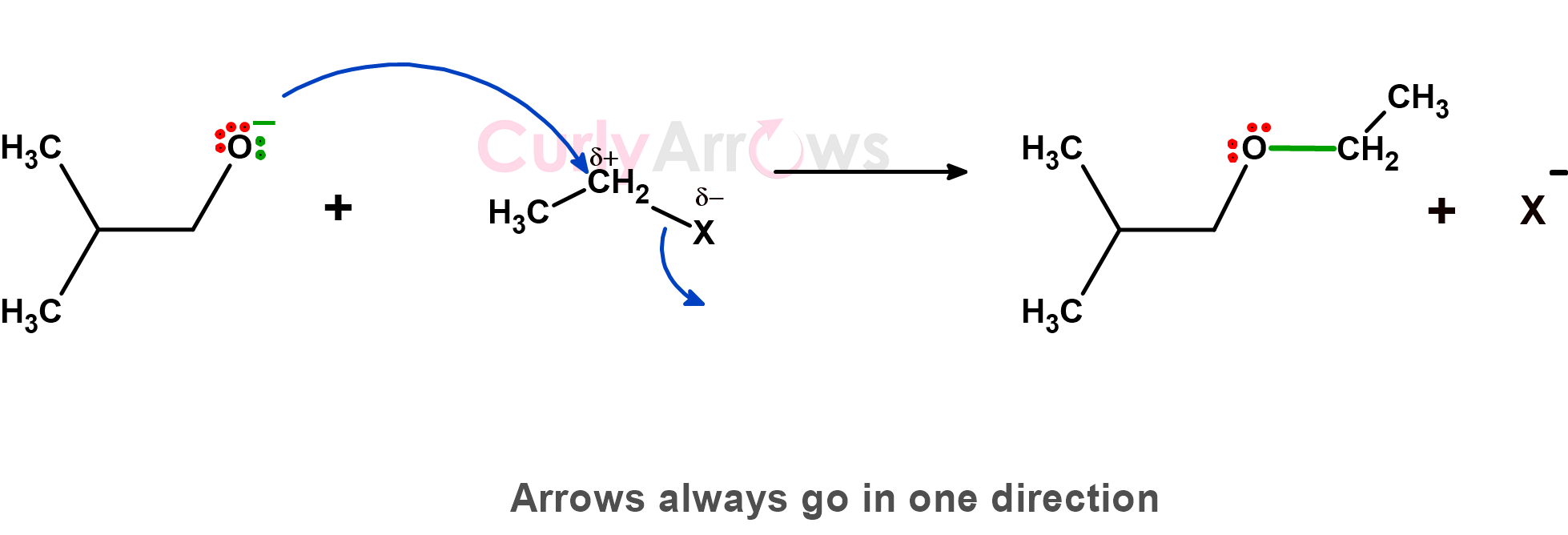
3) The third important commandment is that if multiple arrows are used to depict the path of the electrons, then these arrows should always go in one direction without converging or diverging. They all go either forward or backward.
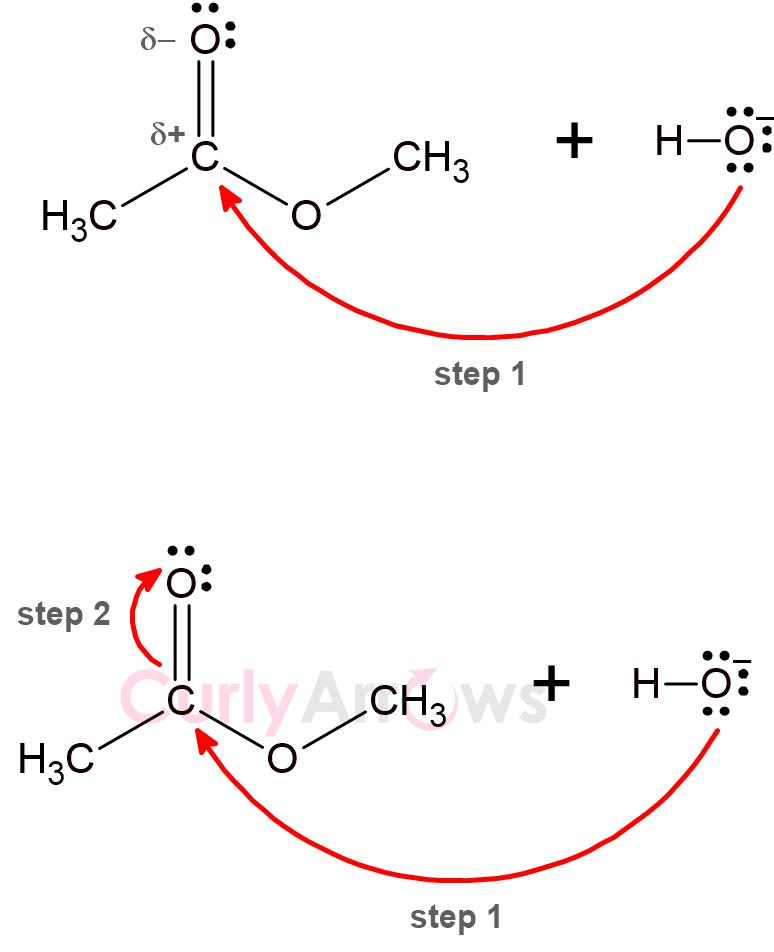
4) The fourth important point is that always preserve an atom’s valency. Every atom on the periodic table has a definite position. This position determines the maximum number of bonds an atom can form.
For example, carbon can form a maximum of four bonds, never five. Therefore, in the example below, as step 1 follows rule number 1, step 2 is pushing the electron density to the electronegative oxygen atom to accommodate a new bond on carbon so that carbon remains tetravalent.
5) Lastly, be mindful of the charges. Charges are preserved.
In the example below, the negative charge on an oxide ion in the starting material is preserved with a bromide ion in the product.

A bond would always break such that the negative charge always resides on the most electronegative atom. For example, in the case of a bond breakage to preserve an atom’s valency (divalent oxygen, tetravalent carbon, and monovalent halogen), the bond breaks such that the two electrons of the bond are donated to the most electronegative atom (the electronegative Bromine as Br-).
A bond would always break in a way that neutralises the positive charge on an electronegative atom.
Take the quiz
Find correct and incorrect methods of drawing curved or curly arrows in reaction mechanisms
Learn how to examine whether the curly arrow mechanism is correct, with examples.
