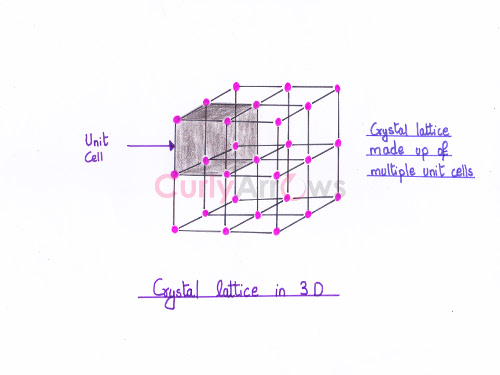
What is an Electrode Potential?
An electrode when in contact with an electrolyte solution of the similar ionic nature (Example, Cu electrode in CuSO4 solution, Zn electrode in ZnSO4 solution) tends to either undergo Oxidation (loss of electrons) or reduction (gain of electrons).
Due to this oxidation or reduction, there develops a charge separation between the metal electrode and its ions in the solution creating a potential difference.