Podcast - Structural Isomerism: The Doppelgangers of Organic Chemistry | CurlyArrows

Inductive Effect | Resonance/Mesomeric Effect |
|---|---|
The sigma (σ) or the covalent bond electrons are pulled or pushed so that the electron density shifts towards the most electronegative atom. | The pie (π) electrons are pulled and pushed through the p-orbitals along the sigma (covalent) bond framework. |
It is a permanent effect. | It is a permanent effect. |
The Atomic number (symbol Z) uniquely identifies an element and is equal to the number of protons present in the nucleus of an atom.
For example, atomic number 6 identifies the Carbon atom with 6 protons in its nucleus. An uncharged Carbon atom will also have 6 electrons in the outer nuclear region, equal to its atomic number.
Learning Objective: To learn about a few common reactions that carbon-centered free radicals undergo in organic chemistry.
Skill Level - Intermediate
Prerequisites:
A decomposition reaction breaks down the substrate into two or more smaller, simpler molecules.
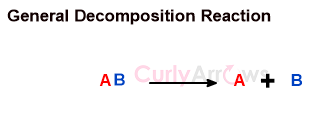
The decomposition reaction is initiated by obtaining energy from heat, light, or electricity sources.
London Dispersion forces are seen in all the atoms and the molecules; however, it is the exclusive force binding nonpolar molecules. Nonpolar molecules are made of two or more atoms, and the electronegativity between two atoms in a covalent bond is less than 0.5. Therefore, no charges are separated to create poles in nonpolar molecules or atoms. Despite not having poles, nonpolar molecules interact using a weak force of attraction known as the London Forces.
Learning Objective: To learn how to predict the stability of a hydrocarbon to form a free radical using the dissociation energy value of the C-H bond.
Skill Level - Intermediate
Prerequisites:
Inorganic chemistry describes oxidation in two ways. The first method is through the loss of electrons, and the second is by the increase in oxidation number.