Around the early 1800s, scientists began to notice the presence of isomerism in all types of compounds; however, at the time, it was referred to using different names.
The term allotrope was used to describe different varieties of an element that existed in different physical forms, such as diamond or graphite for carbon or monoclinic or rhombic for sulfur. Polymorphism was used to explain compounds that occurred in more than one crystalline form, like aragonite and calcite, both crystalline forms of calcium carbonate.
The term isomerism was coined by Berzelius in 1830, recognizing that compounds could share the same molecular formula but differ in their structural arrangement and, consequently, their properties. Berzelius himself had observed that lactic acid from muscle tissue versus that from fermentation exhibited different properties despite having identical chemical formulas.
Since the atoms in a covalently bonded molecule can bond in multiple ways, the term isomerism was slowly being recognised as a phenomenon of covalent compounds containing equal composition and molar masses but showing different properties.
In covalently bonded molecules, the isomerism can be of three types based on the difference in connectivity of atoms, spatial arrangement of various groups of atoms, or based on the rotational form of the molecule.
If the difference between two structures having the same molecular formula and molar masses is due to the different attachment styles of the atoms, then the isomer is constitutional.

If the compounds had the same connectivity of the atoms, however, the groups connecting the atoms differed in their spatial arrangement, then the isomers are stereochemical. Stereochemical isomerism is responsible for the molecules having a permanent shape (or configuration).
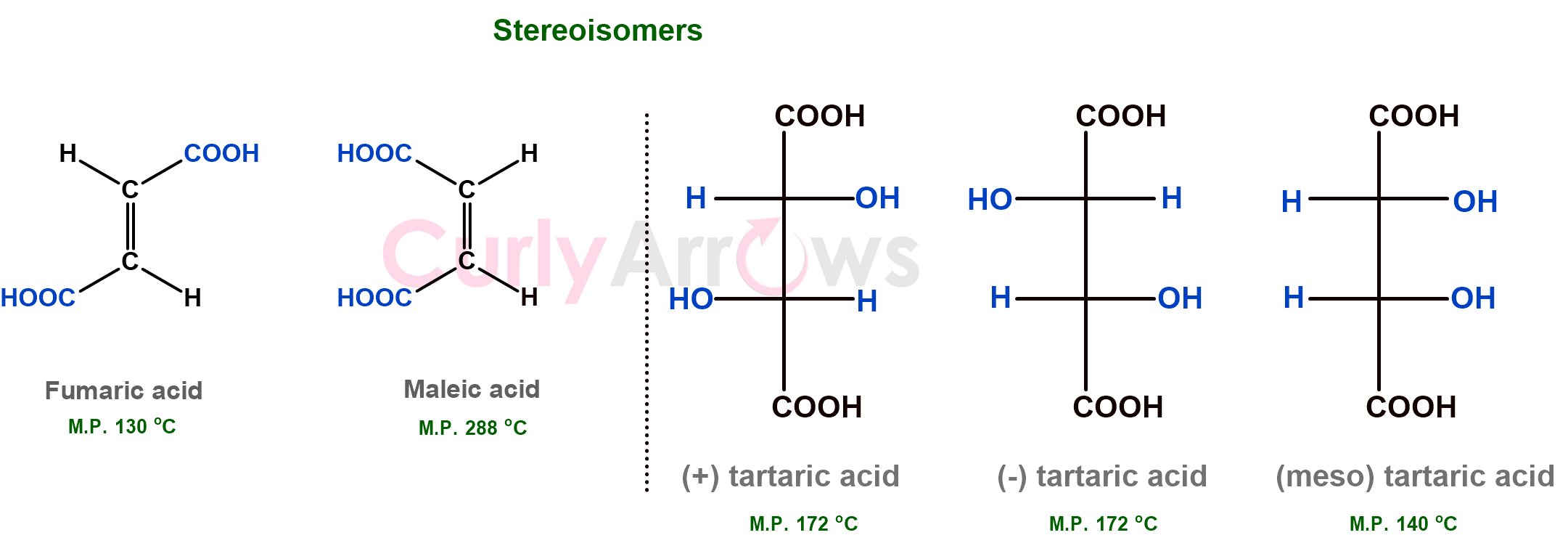
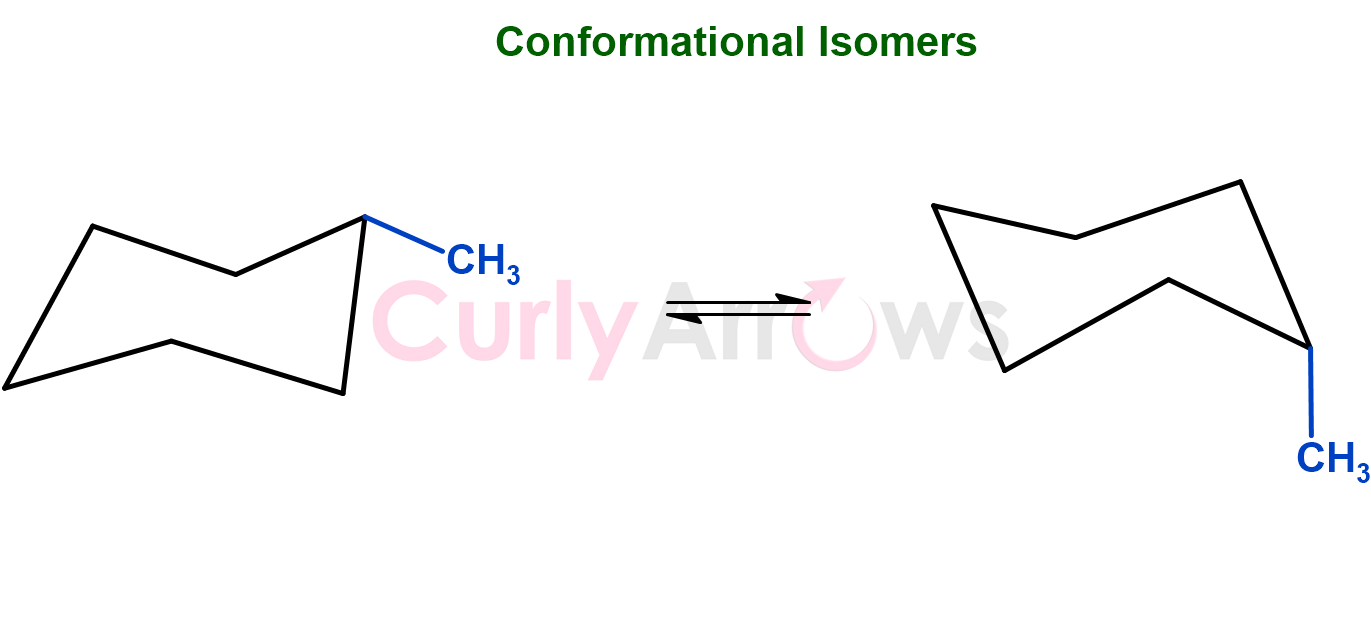
Suppose the compounds had the same constitution and configuration but took temporary shapes (or conformations) due to rotations around single bonds, thereby changing the compounds' geometries. In that case, the isomer is conformational or rotational.
All the above covalent bonds' properties, or their unique ability to exist in different shapes with fixed bond angles, are due to their directional nature. On the other hand, ionic compounds are non-directional, where the force of attraction between the ions acts in all directions. Therefore, they do not show isomerism.
This excerpt is inspired by the chapters Stereoisomerism and Structural Isomerism part of CurlyArrows' Introduction to Organic Chemistry Course. Preview the Book.
Reference: Jacob Berzelius Contribution to Chemistry
Suggested Reading: History of Isomerism (free)
Related Chapter: Preview Structural Isomerism (subscribers only premium tutorial)
